Research Article - Der Pharma Chemica ( 2021) Volume 13, Issue 8
Studies on the performance of micronized Fluconazole for improved health safety and its bioassay against Candida albicans and Aspergillus niger.
Bujji reddy K and Venugopal N. V. S*Venugopal N. V. S, Department of Chemistry, Institute of science, GITAM (Deemed to be University), Visakhapatnam, A. P, India, Email: venu7000@gmail.com
Received: 23-May-2021 Accepted Date: Aug 18, 2021 ; Published: 27-Aug-2021
Abstract
Aspergillus fumigatus and C. albicans are the two most widespread fungal pathogens causing stern insidious infections surrounded by immuno compromised patients. Fluconazole is an antifungal drug of triazole class with incomparable bioavailability. This paper describes the performance of plain fluconazole and micronized fluconazole material for increasing health safety and its biological evaluation by using Aspergillus fumigatus and C. albicans. An active pharma ingredient having antifungal activity was selected and micronized using Microniser to reduce the particle size as low as possible. The product antifungal activity was reviewed between Plain material (heavy particle size, 200 microns) and reduced particle size (micro particles, 2.72 microns). Antifungal drugs with lowest particle size increases efficiency. The Determination of particle size to the micronized material (very fine powder) and its characteristics by laser diffraction and Scanning Electron Microscopy (SEM) and structural identification by Fourier transform Infrared spectroscopy (FT-IR). The efficiency of fluconazole in-terms of antifungal activity of micronized material shows increased efficiency when compared to fluconazole plain material (coarser type particles).
Keywords
Fluconazole, Micronization, Electron microscopy, Bioassay
Introduction
Fluconazole (C13H12F2N6O)(FNE) is an antifungal triazole class drug with exceptional bioavailability. FNE possess low toxicity, and activity against many pathogenic Candida species [1,2]. The IUPAC name is 2-(2, 4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-ol. FNE is applied for treating fungal and yeast infections such as mouth, vagina, throat, esophagus (tube leading from the mouth to the stomach), FNE is functional to treat the various organs like abdomen (area between the chest and waist), and lungs. FNE is exceedingly captivated by the gastrointestinal tract. FNE spreads easily by body fluids. So many unfavorable rejoinders related to the use of FNE are rash, vomiting, nausea, headache, abdominal pain and alopecia in patients undergoing long-standing treatment with a dose of 400 milligrams per day. Prevention of both deep fungal infections in bone marrow transplant populations was the key action of FNE [3,4]. FNE role in an intensive care unit (ICU) leftovers controversial towards patients [5]. In the azole class the fungal confrontation to drugs tends to transpire increasingly over the course of long-standing drug therapy, ensuing in clinical failure in immune negotiated patients [6]. In the TOKU-E's product data sheet [7] the full spectrum of fungal susptibility and confrontation to FNE can be found.
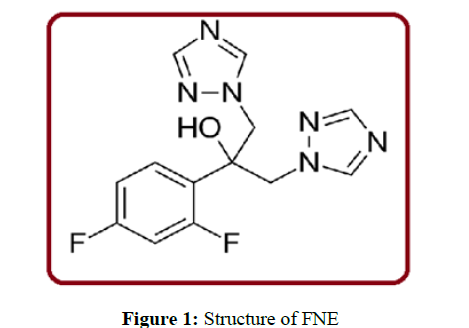
FNE slowdowns the fungal Cytochrome P 450 enzyme 14αdemethylase. As compared to Mammalian the demethylase activity is much less sensitive to FNE. This type of reticence avoid the conversion of lanosterol to ergosterol, an essential component of the fungal cytoplasmic membrane, and subsequent accumulation of 14α-methyl sterols. The structure of FNE was shown in figure 1
Invasive fungal infections are familiar in decisively ill patients expressly those on long-standing mechanical ventilation. Bioassays meet the expense of quite a few practical benefits, such as rapidity, simplicity, and economical. Quite a lot of studies illustrate FNE bioassay using Candida genus [8-14]. This study aimed at to evaluate the performance of micronized FNE to increase health safety and its bioassay by using Candida albicans and Aspergillus niger.
Materials and Methods
Materials
FNE working standard was procured from Granules India limited, Visakhapatnam, and A. P. All other chemicals used were of Analytical reagent grade.
Instrumentation
A laser diffraction technique from Malvern 3000 was used to analyse the particle size of the all the batches. For characterization and identification of molecule Fourier Transform Infra-red Spectroscopy (FTIR) (Bruker) was used. Surface topology was determined by using scanning electron microscopy (SEM) (ZEISS EVO 18).
Preparation of plain material
The white crystalline hygroscopic technical active pharmaceutical ingredient sample was firstly analyzed. The impurities present in the sample are about 0.06%. The residual solvents like methanol, acetone, ethyl acetate, toluene etc., were not detected. The bulk density of the FNE was 0.63g/mL (untapped) and tapped 0.76g/mL. The sieve analysis was conducted and FNE was passed through 20 mesh. The loss on drying was 0.22%w/w.
Preparation of micronized material
The plain material was transferred into dry micronizer equipment with pressure controllers of feeding pressure and micronization pressure. The sample was poured into micronizer hopper with a feed rate of 4 Kg air pressure and 6 Kg micronization pressure with the help of moisture free air. The same procedure was repeated for three cycles to get maximum micronized material.
Test microorganism and continuance
The fungi strains of Candida albicans and Aspergillus niger were used and have been obtained from GVK Bio. Sci, Hyderabad. The pathogenic strains under test was sustained on agar slants (nutrient) and stored at 40C. The slants were preserved in 25 percent glycerol for long period storage.
Antifungal activity preparation (Agarwell diffusion)
In antifungal activity, the potato dextrose agar medium was pendant in 100mL distilled water and heated till complete dissolution. Initially the medium and glass petri-dishes were autoclaved for 20 minutes by the application of 15 Pascal (Pa) pressure. In a laminar flow chamber the medium was poured in to sterile petri-dishes under aseptic conditions.
Results and Discussions
The smaller and finer the particles, the weaker the barriers to dissolution and it lead to dissolution rate. The other benefits of particle size reduction was increased conductivity, smooth surface quality and high quality product function. The surface modification of FNE particles showed remarkable enhancement of antifungal activity.
DLS analysis
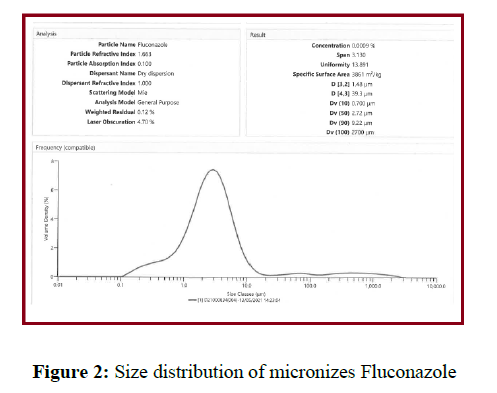
Pade laplace dispersion reveals the diameter of the micronized FNE. Rayleigh scattering, oscillations, Brownian movement and fluorescence exponential decay were the factors of Pade Laplace dispersion. In DLS method a time dependent signal was transformed into the hydro colloidal solution. It results and signifies the exponential decay of the particles. From pade Laplace dispersion graph the transformed signal number of components in the micronized FNE solution can be easily acquired and shown figure 2. One gram of sample is loaded into the sample holder and analysed using laser diffraction particle size analyser using dry accessory. One millilitre of micronized FNE solution was overhanging in 5ml of water. At 25-400C the resultant hydro dispersed suspension was analyzed with DLS. The particle size distribution (shown in figure 2) was emerged 2.72 micrometers.
SEM Analysis
Scanning Electron Microscope (SEM) was selected for knowing the surface topology and size of micronized FNE at diverse magnifications. A drop of micronized FNE solution was taken on the stub and it was air dried and it was subjected to sputtering using sputter coater. In SEM the detection of shape at different scan regions was possible when the area of specimen intermingled with the electron beam produces signals. An electron gun in SEM produces electron and later accelerated through the condenser. The following Scanning Electron Micrographs (shown in figures 3 and 4) illustrates the shape of micronized FNE at a range of magnifications in which micronized FNE particles agglomerated to form clusters. The plain material and micronized material is imaged using scanning electron microscope to know the particle size and shape. The shape of the molecule is cylindrical in plain material and size is ranging from 120 microns to 300 microns. The micronized materials was imaged, the size is around 2 microns and shapeless, since the powder is fine.
FT-IR analysis
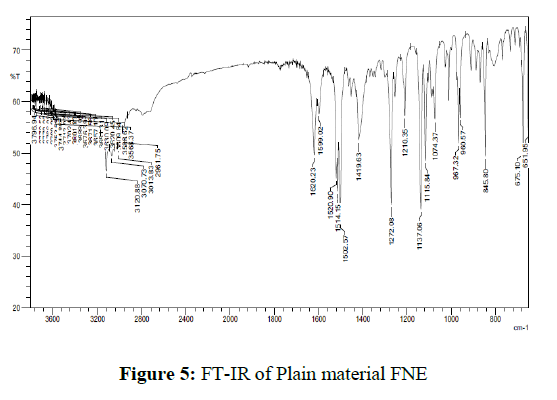
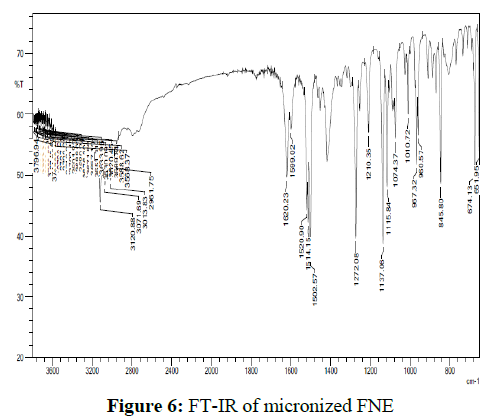
A small amount of micronized FNE in KBr pellets in transmittance mode was processed for categorization of the functional groups. FT-IR analyses were conceded out to identify the possible micronized FNE. 2 mg of sample was mixed with 200 mg of KBr and grounded to make a transparent disc and scanned using FT-IR spectrophotometer and the spectrum is recorded. No change in the chemical structure of micronized FNE as palpable from the presence of peaks due to same functional group was observed from FT-IR studies. Presence of peak at 1620cm-1 in both spectra (C=N) make obvious that the presence of triazole ring. The vibrations of the diverse functional groups present in the molecule could be ascribed to a broad band due to hydrogen bonded O–H stretching vibrations in the range of 3,600–2,500 cm-1.1520 cm-1and 1419 cm-1 bands due to triazole ring stretch. 960 cm-1 and 845 cm-1 reveals C-H triazole ring. The FT-IR spectra of plain material and micronized FNE were shown in figures 5 and 6.
Application of micronized FNE on Candida albicans and Aspergillus Niger
Aspergillus fumigatus and C albicans (figure 7) are the two most common fungal pathogens causing severe invasive infections among immuno compromised patients
Cylinder-Plate or Cup-Plate Method (Agar Diffusion method)
Dissolved one gram of Fluconazole sample in 0.1N HCl inoculated with a beforehand liquefied medium apposite to the assay with the indispensable quantity of suspension of the micro-organism Candida albicans and Aspergillus niger. Added the suspension to the media (sabouraud Dextrose Agar) and immediately pour the inoculated media into the Petri dishes (or) large rectangular plates to give a depth of 3 to 4mm. The layers of medium are uniform in thickness by placing the dishes or plates on a level surface. All prepared plates were stored carefully so that no significant growth or death of the test organism occurs before the plates are used and that the surfaces of the agar layer dry at the time of use. Known concentration of the sample that is 1mg/mL of fluconazole is prepared. 3 to 4 mm holes are made on the separate agar plates for two batches that is 001(normal) and 004 (Micronized) for both above mentioned organisms with the help of sterile borer. Known concentration of both the products that is 001 and 004 1mg/ml is added into the holes for respective organisms. Same volume of solution added to each cylinder. Under Laminar air flow t the plates kept for one to four hours duration at room temperature. Incubated them for about 3 to 5 days at 20 to 250c. Of the Circular inhibition diameter were accurately measured. The anti-fungal activity of plain and micronized FNW was shown in figure 8.
The results obtained about the effectiveness of FNE on Candida albicans and Aspergillus niger were given in table 1.
| Organism | Zone of Inhibition for Fluconazole( Micronized) | Zone of Inhibition for Fluconazole (Normal) | ||||
|---|---|---|---|---|---|---|
| Study Plate -1,cm | Study plate-2,cm | Average cm |
Study Plate -1,cm |
Study Plate -2,cm |
Average cm |
|
| Aspergillus niger | 1.0 | 0.8 | 0.9 | 0 .3 | 0 .2 | 0.4 |
| Candida albicans | 2.0 | 2.0 | 2.0 | 1.6 | 1.6 cm | 1.6 |
Conclusion
Aspergillus fumigatus and C albicans are the two most common fungal pathogens causing severe invasive infections among immuno compromised patients. FNE is a fluorine-substituted, bis-triazole antifungal agent. In the mechanism of FNE involves the interruption of the conversion of lanosterol to ergosterol via binding to fungal cytochrome P-450 and succeeding distraction of fungal membranes. From the above study, it is proved that the efficiency of fluconazole in-terms of antifungal activity of micronized material shows increased efficiency when compared to fluconazole plain material(coarser type particles).
Conflict of Interests
The author declared that, having no conflict of interests.
Acknowledgements
We are thankful to the management, faculty of chemistry, GITAM University Visakhapatnam, AndhraPradesh, India for their support and encouragement given to us.
References
- Saag MS and Dismukes WE. Antimicrob Agents Chemother. 1988, 32: p. 1–8.
- Goodman JL, Winston DJ, Greenfield RA et al., N Engl J Med. 1992, 326: p. 845–51.
- Winston DJ, Chandrasekar PH, Lazarus HM et al., Ann Intern Med 1993, 118: p. 495–503.
- Edwards JE, Bodey LGP, Bowden RA et al., Eur J Clin Microbiol Infect Dis. 1997, 25: p. 43–59.
- Slotman GJ and Burchard KW. Arch Surg. 1987, 122: p. 147–51.
- Amantea MA, Stevens DA and Bennett JE, Antimicrob Agents Chemother. 1991, 35: p. 846.
- Mendes FES, Oliveira LVN, Fri ES et al., Eur J Clin Microbiol Infect Dis. 2010, 29: 1525.
- Hurtado FK, Souza MJ, Melo J de et al., LatAm J Pharm. 2008, 27: p. 224.
- Marchetti O, Majcherczyk Paul A, Glauser Michel P et al., Antimicrob Agents Chemother. 2001, 45: p. 696.
- Tan AL and Chan KS. Ann Acad Med Singapore, 2008, 37: p. 841.
- Hawser S and Islam K. J Antimicrob Chemother. 1999, 43: p. 411.
- Pelletier R, Loranger L, Marcotte H et al., J Med Microbiol. 2002, 51: p. 479.
- Linares MJ, Charriel G, Solis F et al., J Clin Microbiol, 2004, 2: p. 899.
- Rewak-Soroczynska J, Sobierajska P, Targonska S et al., Int. J. Mol. Sci, 2021, 22: p. 3112.