Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 2
Spectrometric Determination of Amitriptyline in Real Samples after Separation by Two Liquid-phase Microextraction Method
Farideh Mofazzeli
Abstract
The main aim of this research is using the preconcentration and sample preparation method, based on directly suspended drop microextraction method of amitriptyline prior to UV-vis spectrophotometer. Here, the target compound was extracted from an aqueous sample solution (donor phase) into an organic solvent drop (acceptor phase), which was suspended in the sample solution without using a microsyringe needle as a supporting device. Several important parameters which influence the extraction efficiency such as the kind of extracting solvent, extraction time, volumes of the sample solution and organic extraction solvent, stirring rate, pH of the donor phase and salt effect were investigated. Under the optimal conditions the enrichment factor was 25. By plotting absorbance versus concentration of the analyte in the standard solutions, calibration curve was obtained with correlation coefficient of 0.9931. The linearity of the method has been investigated between the ranges of 0.04-1 μg mL-1. The Limit of Detection (LOD) of the method which calculated theoretically was 0.013 μg mL-1. The precision or repeatability of the method is based on the average Relative Standard Deviation (RSD%) is 4.6, for three different concentrations of the analyte. Finally, the proposed method was applied for the determination of amitriptyline in urine and environmental water samples and the reasonable relative recoveries were obtained.
Keywords
Directly suspended drop microextraction (DSME), Amitriptyline, UV-vis spectrophotometer
Introduction
One of the most widespread and common mental disorders in the world is depression that is one of the serious global economic problems because the patients often lose the ability of working and also, it may ultimately result in suicide [1,2]. The depression treatment includes pharmacotherapy with medicines as well as various forms of psychotherapy. One of the largest groups of drugs for treatment of psychiatric disorders is the Tricyclic Antidepressants (TCAs) which are widely used for treatment of depression [3]. Amitriptyline is one of the TCAs has been used to treat endogenous depression, phobic states, panic attacks, neuropathic pain states, and pediatric enuresis.
Sample preparation is still the most important challenge for the analysis of different compounds from various complex matrices, especially real samples. Conventional Liquid–Liquid Extraction (LLE) and Solid Phase Extraction (SPE) have been usually applied as the useful sample preparation methods for determination of drug for many decades [4,5]. However, both methods have certain drawbacks. LLE is a time consuming and tedious procedure and needs very large amounts of the high-purity, expensive and also hazardous organic solvents. In SPE methods often the artifacts introduce into the sample solution; therefore, the limitation for pH ranges of this solution is very important factor. On the other hand, may require lengthy processing such as washing, conditioning, eluting and solvent evaporation [6]. During the last decade, new modern sample preparation methods with respect to simplification, miniaturization, and minimization of the organic solvent usage have been developed. Stir-Bar Sorptive Extraction (SBSE), Solid Phase Microextraction (SPME) and Liquid Phase Microextraction (LPME) are miniaturized techniques; introduced for these purposes [7-14]. SPME and SBSE are simple and solventless methods. However, the major disadvantages of SPME are SPME fibers are still comparatively expensive and the polymer coatings are fragile and have limited lifetime [12].
LPME can be divided into three broad categories: Single Drop Micro Extraction (SDME), Hollow Fiber Based Liquid Phase Microextraction (HF-LPME) and Dispersive Liquid-Liquid Microextraction (DLLME) [14-20]. Single drop microextraction is a mode of LPME that provides analyte extraction in a few microliters of an organic solvent [14-18]. SDME avoids some problems of the Solid Phase Microextraction (SPME) method such as sample carry-over and fiber degradation. It is also quick, inexpensive and uses very simple equipments. In the SDME technique, a microdrop of an organic solvent is immersed in a stirred aqueous sample solution [15-18]. In recent years, Lu and co-workers [21] developed Directly Suspended Droplet Microextraction Method (DSDME) as a new sampling method of SDME. In this method, a stirring bar is placed at the bottom of a vial containing an aqueous sample and rotated at a speed required to cause a gentle vortex. If a small volume of an immiscible organic solvent is added to the surface of the aqueous solution, the vortex results in the formation of a single droplet at or near the centre of rotation. The droplet itself may also rotate on the surface of the aqueous phase, increasing mass transfer. Compared with the other LPME techniques based on drop systems (e.g., SDME), it provides more flexibility in the choice of the operational parameters, especially the amount of the organic solvent and the stirring frequency. The possibility of applying larger volumes of organic solvents in this method also makes it a useful technique to match with HPLC and UV-vis spectrophotometer [13,21].
In the present work, we used DSDME method for extraction and preconcentration of the target compound (amitriptyline) in real biological and water samples (urine and groundwater) prior to UV-vis spectrophotometer.
Materials and Methods
Chemicals and materials
Amitriptyline with purity of >99% was kindly supplied from Darou Pakhsh Co. (Tehran, Iran). Analytical reagents grade such as: 1-octanol, n-hexane, n-heptane and methanol (high purity) were purchased from Merck (Darmstadt, Germany). All the other used chemicals such as, acetone, NaCl, HCl and NaOH were purchased from Merck (Darmstadt, Germany) and used without further purification.
Preparation of samples
Stock solution of amitriptyline (1000 μg mL−1) was prepared by dissolving calculated amount of the drug in methanol and stored protected from light in refrigerator at 4ºC. Fresh working solutions in various concentrations were prepared daily by diluting the appropriate amount of stock solution in distilled water and the solution pH was adjusted.
Apparatus
Spectrophotometric measurements were carried out using a UV-vis Unicode 2100 spectrophotometer (USA) equipped with a quartz microcell of appropriate path length with internal volume of 300 μL. The IKA heating magnetic stirrer (50-2500 rpm, Germany) was used for agitation of the sample solutions in the microextraction procedure. A 500 μL HPLC microsyringe with a flat needle was used for the sample injectors of the microextraction equipments.
Directly suspended droplet microextraction
The experimental setup of DSDME is illustrated in Figure 1. A cylindrical sample cell with a PTFE coated stirring bar was placed on a heating-magnetic stirrer. Then, a volume of 5 mL aqueous sample solution containing 1 μg mL−1 of amitriptyline was transferred into the vial as donor phase. The magnetic stirrer was turned on and adjusted to desired stirring speed. A trifle of n-hexane (180 μL, acceptor phase) was dripped on the centre of the aqueous sample surface carefully with a HPLC microsyringe and the mixture was agitated for 10 min at 1800 rpm. After this time, the acceptor phase was retracted into the microsyringe and transferred into the quartz microcell and introduced to the spectrophotometer for measuring the absorbance at 240 nm.
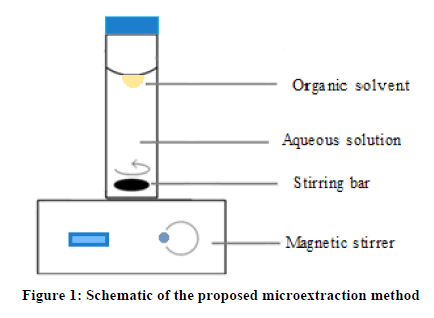
Conclusion
The present work describes the possibility of using DSDME method in the extraction of amitriptyline from water samples prior to UV-vis spectrophotometer by utilizing a simple, rapid and cheap extraction device. In this method, contrary to the ordinary single drop liquid phase microextraction technique, an organic large drop is freely suspended without using a microsyring as supporting device. This large drop causes an increasing in mass transfer process and decreasing in equilibrium time. Compared to the most conventional extraction procedures, this extraction technique requires a very little aqueous sample solution and very little expensive and toxic organic extractants. On the other hand, this method is very fast, easy and simple. Using this technique, the analyte can be extracted from real biological and water samples quantitatively with a good linearity and reasonable relative recoveries.
Acknowledgment
The authors would like to acknowledge the Quchan branch, Islamic Azad University, Iran, for the financial support of this work.
References
[1] http://www.who.int/mediacentre/news/notes/en/
[2] W.Z. Potter, L.E. Hollister, McGraw-Hill, NY, USA, 2007.
[3] M. Furlanut, P. Benetello, E. Spina, Clin. Pharmacokinet., 1993, 24, 301.
[4] J. Wang, M. Bonakdar, C. Morgan, Anal. Chem., 1986, 58, 1024.
[5] R. Theurillat, W. Thormann, J. Pharm. Biomed. Anal., 1998, 18, 751.
[6] T. Gunnar, S. Mykkänen, K. Ariniemi, P. Lillsunde, J. Chromatogr. B., 2004, 806, 205.
[7] P. Sandra, B. Tienpont, J. Vercammen, A. Tredoux, T. Sandra, F. David, J. Chromatogr. A., 2001, 928, 117.
[8] B. Kolahgar, A. Hoffmann, A.C. Heiden, J. Chromatogr. A., 2002, 963, 225.
[9] E. Psillakis, N. Kalogerakis, Trends Anal. Chem., 2003, 22(10), 565.
[10] S. Pedersen-Bjergaard, K.E. Rasmussen, Anal. Chem., 1999, 71, 2650.
[11] J.M. Kokosa, Trends Anal. Chem., 2013, 43, 2.
[12] C.L. Arthur, J. Pawliszyn, Anal. Chem., 1990, 62, 2145.
[13] A. Sarafraz-Yazdi, A. Amiri, Trends. Anal. Chem., 2010, 29, 1.
[14] A. Sarafraz Yazdia, F. Mofazzeli, Chromatographia., 2010, 72, 867.
[15] M.A. Jeannot, F.F. Cantwell, Anal. Chem., 1996, 68, 2236.
[16] E. Psillakis, N. Kalogerakis, Trends. Anal. Chem., 2002, 21, 53.
[17] A. Sarafraz Yazdi, F. Mofazzeli, Z. Es'haghi, Chromatographia., 2008, 67, 49.
[18] M.A. Jeannot, A. Przyjazny, J.M. Kokosa, J. Chromatogr. A., 2010, 1217(16), 2326.
[19] A. SarafrazYazdi, F. Mofazzeli, Z. Es'haghi, Talanta, 2009, 79, 472.
[20] M. Rezaee, Y. Assadi, M.R.M. Hosseini, E. Aghaee, F. Ahmadi, S. Berijani, J. Chromatogr. A., 2006, 1116, 1.
[21] Y.C. Lu, Q. Lin, G.S. Luo, Y.Y. Dai, Anal. Chim. Act., 2006, 566, 259.
[22] A. Sarafraz Yazdia, F. Mofazzeli, Z. Es’haghi, J. Chromatogr. A., 2009, 1216, 5086.
[23] H. Lord, J. Pawliszyn, J. Chromatogr. A., 2000, 902, 17.
[24] Y. Wang, Y.C. Kwok, Y. He, H.K. Lee, Anal. Chem., 1998, 70, 4610.



