Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 1
Impact of Sub Fertile Males BMI on Sperm Parameters and Some Seminal Antioxidants
Al-Zayadi SM1, Al-Murshidi SY2* and Al-Katib SR1
1Department of Physiology, College of Medicine, University of Kufa, Iraq
2Department of Urology, College of Medicine, University of Kufa, Iraq
- Corresponding Author:
- Al-Murshidi SY
Department of Urology, College of Medicine, University of Kufa, Iraq
Abstract
Ninety subfertile couples were involved in the present study during their attendance at fertility clinic at Al-Sadder teaching hospital in Al- Najaf/Iraq throughout a period from March 2016 to January 2017. Details history and physical examination were done for every subject participated in this study. Body Mass Index (BMI) was measured for all male participants that divided according to it into three categories including normal weight, overweight and obese males. The objective of this study was to investigate whether increased male BMI has any impact on sperm parameters or on seminal antioxidants. Main results of this study revealed that there was a significant differences (p<0.05) in sperm parameters and in the antioxidants levels among different male BMI categories. With increasing male BMI there was a decrease in sperm parameters and in seminal antioxidants levels. It was concluded that increased male BMI have a negative impact on different sperm parameters including (concentration, motility, morphology and volume) and on the levels of seminal antioxidants including (Glutathione, catalase and Super Oxide Dismutase (SOD)).
Keywords
Antioxidants, BMI, Morphology, Glutathione
Introduction
Infertility could be defined as the couples inability to achieve spontaneous conception after a period of one year or more of regular, unprotected sexual relationship [1]. Infertility is a growing concern affecting globally up to 15% of couples trying to conceive, and it is reported that male factors are involved in about 50% of infertile relationships. Fortunately, progresses in the Assistive Reproducing Technologies (ART) have assisted many subfertile couples to achieve conception [2]. But with modern life and changing lifestyles, favoring the development of obesity there’s been an alarming increase in subfertility and new researches have suggested a potential link between male subfertility and obesity [3].
Obesity could be defined as a health distress where extra body fat collects in the body to the degree that this collection of fat might affects health adversely. The most common assessment of body fatness is Body Mass Index (BMI) which could be calculated by the division of person’s weight measured in kilograms by their height in square meters [4]. Subject could be considered as being normal if their BMI is, 18.5-24.9 kg/m2; with the range 25-29.9 kg/m2 as overweight and BMI of 30 kg/m2 or greater as obese [5]. Extra fat is not only associated with increased danger of chronic diseases, but has also been revealed to increased danger of reproductive problems [6] and the mechanisms that responsible for the influence of obesity on man fertility are mostly dubious and uncertain [7]. Elevation in the number or size of fat cells due to obesity can results in two types of changes, physical and hormonal. Physical type of changes may involve; rise in the occurrence of sleep apnea, rise in scrotal temperature, and rise in erectile dysfunction, while hormonal one may involve an elevation in estrogen, leptin hormone, and insulin levels, also a decline in testosterone, inhibin-B and Sex Hormone Binding Globulin (SHBG) levels. Also the production of Gonadotropin Releasing Hormone (GnRH) is impaired with increasing body fat [8]. These hormone profile changes that results from dysregulation of hypothalamic pituitary gonadal (HPG) axis may impair spermatogenesis process and other features of male reproduction and in turn, contribute to azoospermia, oligozoospermia, increase in the DNA Fragmentation Index (DFI) and a reduction in the semen volume [9].
Obesity also can affect male fertility by Oxidative Stress (OS) that initiate damage to spermatozoa [10]. Sperms and seminal plasma contain plentiful antioxidants, but if the generation of Reactive Oxygen Species (ROS) by the immature sperms or by the leucocytes of the semen is excessive these defensive system could be overcome, which may lead to damage of the sperm DNA [11]. This state is known as oxidative stress and can lead to severe cellular damage [12]. Obesity may encourage systemic state of oxidative stress [13], since obese persons are in a longlasting state of inflammation due to fat tissue production of proinflammatory cytokines like Tumor Necrosis Factor Alpha (TNFα) and Interleukin 6 (IL-6), which can result in the stimulation of leucocyte for the production of ROS and also the leptin production by fat tissue has been stated to increase the oxidative metabolism of the sperms [14].
Materials and Methods
Patients
A total of ninety subfertile couples who approach the clinic of fertility center at Al-Sadder teaching hospital in Al-Najaf/Iraq for their inability to conceive were included in this study, throughout a period from March 2016 to January 2017. Details history and physical examination was done for every subject participated in this study. Subfertile males with genetical, anatomical defects or past history of infections such as measles, mumps, pneumonia, tuberculosis, sexually transmitted diseases or exposure to radiation, heat or any surgical procedures were excluded from the study.
Body Mass Index (BMI) calculation
Weight and height of the subfertile male was calculated using sensitive balance for weight and height after removing excessive clothing (e.g. Shoes, jackets, coats, and sweaters). For measuring height, male was asked to stand erect with his back to the wall and relaxed shoulders at the sides and looking straight ahead. Patient's height (meters) and weight (kilograms) were recorded. BMI was calculated as the body weight in (kilograms) divide by the square of height in (meters). Participants were classified into three categories according to the following published BMI ranges: BMI of 18.5-24.99 kg/m2 was classified as normal, 25-29.99 kg/m2 as overweight, 30 kg/m2 and over as obese [5].
Semen collection and analysis
All samples were collected from the participants by masturbation onsite, and analyzed in reference to WHO guidelines. participants were requested to refrain from sex for (2–7) days before the time of collection. They received a dry, clean, pre weighed and biologically inert container that had been labeled clearly with the patient’s full name. Samples that collected were liquefied for 30 min at 37°C, after that the liquefied semen was mixed carefully for few seconds, and then investigated by macroscopical and microscopical examinations within not more than sixty minutes of collection according to the WHO guideline [1].
According to the results of the Seminal Fluid Analysis (SFA) we categorized our patients into two groups: Normozoospermic patients for those who have normal semen parameters (sperm concentration > 20*106 sperms/ml, and have not less than 32% of sperms with progressive motility, with normal sperm morphology). Asthenozoospermic patients for those who have normal sperm concentration and morphology, but have less than 32% of sperms with progressive motility.
After centrifugation process, we obtain the seminal plasma that divided into many abendrof tubes and stored frozen until the time of biochemical tests for the measurement of the levels of some seminal antioxidants (Glutathione, Superoxide Dismutase (SOD) and catalase) according to available kits.
Results
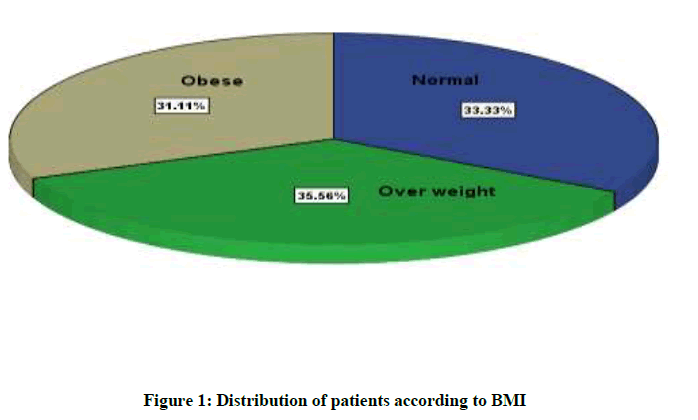
In this study, we evaluate the influences of male BMI on sperm parameters (Sperm concentration, progressive sperm motility percent, normal sperm morphology percent and semen volume) and on some seminal antioxidants (Glutathione, SOD and catalase). Figure 1 shows the classification of the subfertile males according to their BMI. Where it shows highest percent for overweight patients (36.56%) and lowest for obese patients (31.11%), while the percent of normal weight patients was (33.33%).
Table 1 shows significant difference in semen parameters among BMI categories. With increased BMI there was a decrease in these parameters. Highest level of sperm concentration seen in normal weight patients (73.63 ± 32.58) and lowest level in obese patients (40.82 ± 21.09) while in overweight patients it was (54.96 ± 25.64) p < 0.001. Highest level of progressive sperm motility percent seen in normal weight patients (56.66 ± 27.42) and lowest level in obese patients (36.41 ± 17.92) while in overweight patients it was (47 ± 19.52) p=0.003. Regarding normal sperm morphology percent highest level seen in normal weight patients (60.33 ± 18.84) and lowest level in obese patients (36.96 ± 14.61) while in overweight patients it was (49.06 ± 20.41) p < 0.001. Highest level of semen volume seen in normal weight patients
| Parameter | Normal weight (n=30) | Overweight (n=32) | Obese (n=28) | P value |
|---|---|---|---|---|
| Concentration (million/ml) | 73.63 ± 32.58abc | 54.96 ± 25.64abc | 40.82 ± 21.09abc | <0.001 |
| Progressive motility (%) | 56.66 ± 27.42ac | 47 ± 19.52 | 36.41 ± 17.92ac | 0.003 |
| Morphology (%) | 60.33 ± 18.84abc | 49.06 ± 20.41abc | 36.96 ± 14.61abc | <0.000 |
| Volume (ml) | 3.58 ± 0.54abc | 2.67 ± 0.72abc | 2.21 ± 0.82abc | <0.001 |
abc: Significant between normal weight, overweight and obese; ac: Significant between normal weight and obese
Table 1: Semen parameters in relation to BMI
Table 2 shows significant difference in ROS level among different BMI categories. Highest ROS level seen in obese patients (6.34 ± 2.23) and lowest in normal weight patients (2.55 ± 1.666) while in overweight patients it was (4.02 ± 1.648) p<0.001. Highest glutathione level seen in normal weight patients (251.09 ± 50.17) followed by that of overweight patients (233.92 ± 49.6) and the lowest level seen in obese patients (165.31 ± 91.52) p<0.001. So there was a significant differences in the level of glutathione among different BMI categories Also there was a significant differences in the level of both catalase and SOD among the different BMI categories. Highest catalase and SOD value seen in normal weight patients (4.22 ± 2.07) and (926.93 ± 312.7) respectively followed by that of overweight patients (3.27± 1.76) and (666.53 ± 256.34) and the lowest level seen in obese patients (2.62 ± 1.08) and (535.34 ± 272.49) p=0.002 for catalase and p <0.001 for SOD. (3.85 ± 0.54) and lowest level in obese patients (2.21 ± 0.82) while in overweight patients it was (2.67 ± 0.72) p=0.001.
| ROS and antioxidants | Normal weight (n=30) | Overweight (n=32) | Obese (n=28) | P value |
|---|---|---|---|---|
| ROS (ng/ml) | 2.55 ± 1.666abc | 4.02 ± 1.648abc | 6.34 ± 2.234abc | <0.001 |
| Glutathione (µg/ml) | 251.09 ± 50.17a | 233.92 ± 49.6b | 165.31 ± 91.52ab | <0.001 |
| Catalase (ng/ml) | 4.22 ± 2.07ac | 3.27 ± 1.76a | 2.62 ± 1.08c | 0.002 |
| SOD (pg/ml) | 926.93 ± 312.7ac | 666.53 ± 256.26a | 535.34 ± 272.49c | <0.001 |
Similar letters means significant
Table 2: ROS and some antioxidant in relation to BMI
Discussion
Obesity has become widespread not only in high-income nations, but increasingly so in all others, the numbers of overweight persons are rapidly on the increase, resulting in a global obesity epidemic [15]. Our study shows significant difference in all semen parameters (sperm concentration, progressive sperm motility percent, normal sperm morphology percent and semen volume) among different BMI categories, with increase BMI there was a decrease in these parameters.
Jensen et al. [16] measured the association between BMI and semen quality, they found that there was some significant correlations between sperm count and BMI, patients with normal BMI shows greater sperm concentration as well as a greater total sperm count and a lower percent of abnormal sperms. Hammoud et al. [10] found that with increasing male BMI the progressive motile sperm count and sperm concentration was decreased. According to Hammiche et al. [17] there was a negative impact of increased male BMI on sperm count and motility, Macdonald et al. [18], found negative impact on sperm morphology only, while Chavarro et al. [19] reported that with increasing male BMI the ejaculate volume and the sperm count decrease. Sharma et al. [10] showed that there is an association between male BMI and sperm parameters alteration, and he found that obese males are three times more likely to show a drop in semen quality than males of a normal BMI.
The explanation of the correlation between increased male BMI and the production of sperms is undefined and likely complicated. In the light of our results, it seems that man’s BMI is inversely related to the concentration of androgens in the body and to SHBG-levels, A decrease in the level of SHBG results in a decreased concentration of free testosterone in the blood [20]. The concentration of inhibin B decreases with an increase in BMI but this change is not accompanied by a compensatory increase in FSH. However, it has been shown that an increase in BMI in the man is positively related to estrogen levels. This relationship arises as a result of increasing conversion by aromatization of androgens to estrogens in the peripheral fat tissue in overweight and obese individuals as compared with individuals of normal weight. High estrogen levels have a damaging effect on endogenous secretion of gonadotropin because they interfere with GnRH-pulsatility. Overweight men may, as a result, be affected by hormonal changes similar to those associated with hypogonadotropic hypogonadism, which is to say low gonadotropinand testosterone concentrations. These hormonal changes that occur in overweight men are particularly evident in abdominal fatty tissue [21].
Our study also revealed that there was a significant differences in the levels of antioxidants glutathione, catalase and SOD among different BMI categories. Highest levels seen in normal weight patients followed by that of overweight patients and the lowest level seen in obese patients. One of the significant elements of the seminal plasma is the SOD. Superoxide anion is an important antioxidant and has an important scavenging capacity, several investigators have described reductions of the activity of SOD in the semen of subfertile men leading to lipid peroxidation state [22]. The results of the current study are consistent with the studies by Murawski et al. [23] and Nissen et al. [24] which noted a positive relationship between seminal plasma SOD activity and the quality of semen parameters of sperm concentration and overall motility by prevention of lipid peroxidation, and they found that there was a significant reduction in the activity of semen SOD in subfertile males, in comparison with normozoospermic one. Also our study shows that catalase activity was significantly reduced, and this result agreed with the results of study by Sanocka et al. [25] which found that catalase activity was lower in seminal plasma of asthenozoospermic patients compared to normal one, and it was confirmed by statistically significant relationship between the enzyme activity and the number of sperms. catalase enzyme is capable to reduce hydrogen peroxide, in addition it activate nitric oxide that induce sperm capacitation.
Antioxidants are molecules that defend biological biomolecules against OS, fewer availability of antioxidants resulting in a state of oxidative stress, since plasma membrane of the sperm contain high quantity of lipids in the form of Polyunsaturated Fatty Acids (PUFA), ROS assaults PUFA in the plasma membrane resulting in lipid peroxidation, antioxidants act to prevent spontaneous oxygen toxicity and lipid peroxidation of sperms as well as prevent premature hyper activation and capacitation that may occur. Different studies revealed that lipid peroxidation influences the sperm count, motility, morphology and also associated with reduced sperm quality [26,27].
Conclusion
In the light of our results it seems that, bad quality of sperm parameters that was reported for the high BMI males may be the result of the imbalance between ROS production and antioxidants scavenging activities. The antioxidants levels were significantly reduced and this reduction in the scavenging capacity would lead to OS that leading to decreased sperm concentration and motility.
References
- World Health Organization (WHO): Laboratory Manual for the Examination and Processing of Human Semen 5th Edi., Department of Reproductive Health and Research, 2010.
- M. Levy, Reproductive Physiology, 1st Edi., Britannica Educational Publishing In association with Rosen Educational Services, LLC 29 East 21st Street, New York, 10010, 2011.
- N. Stefan, H.U. H€aring, F.B. Hu, M.B. Schulze, Lancet Diabetes Endocrinol., 2013, 2, 152-162.
- K. Esposito, D. Giugliano, Int. J. Impot. Res., 2005, 17, 391-398.
- World Health Organization, Office of Health Communications and Public Relations, Obesity and overweight. Geneva: WHO, 2006.
- P. Catalano, Obstet. Gynecol., 2007, 109, 419-433.
- S.E. Wozniak, L.L. Gee, M.S. Wachtel, E.E. Frezza, Dig. Dis. Sci., 2009, 54, 1847-1856.
- M. Sallmen, D.P. Sandler, J.A. Hoppin, A. Blair, D.D. Baird, Epidemiology., 2006, 17, 520-523.
- A.O. Hammoud, N. Wilde, M. Gibson, A. Parks, D.T. Carrell, Fertil. Steril., 2008b, 90, 2222-2225.
- R. Sharma, K.R. Biedenharn, J.M. Fedor, A. Agarwal, Reprod. Biol. Endocrinol., 2013, 11, 66.
- R.J. Aitken, G.N. De Iuliis, Reprod. Biomed. Online., 2007, 14, 727-733.
- S. Kamarzaman, A. Abdul Wahab, S. Abdul Rahman, Global Veterinaria., 2014, 12(1), 80-89.
- A.I. Ruperez, A. Gil, C.M. Aguilera, Int. J. Mol. Sci., 2014, 15, 3118-3144.
- F. Lampiao, S.S. du Plessis, Asian J. Androl., 2008, 10, 799-807.
- World Health Organization (WHO): Technical Report: Diet, Nutrition and the Prevention of Chronic Disease, 2013.
- T.K. Jensen, A.M. Andersson, N. Jørgensen, A.G. Andersen, E. Carlsen, J.H. Petersen, Fertil. Steril., 2004, 82, 863-870.
- F. Hammiche, J.S. Laven, J.M. Twigt, W.P. Boellaard, E.A. Steegers, R.P. Steegers-Theunissen, Hum Reprod., 2004, 27, 2365-2372.
- A.A. Macdonald, A.W. Stewart, C.M. Farquhar, Hum. Reprod., 2013, 28, 3178-3187.
- J.E. Chavarro, T.L. Toth, D.L. Wright, J.D. Meeker, R. Hauser, Fertil Steril., 2010, 93, 2222-2231.
- B.L. Wajchenberg, Endocr Rev., 2000, 21, 697-738.
- R. Pasquali, Fertil Steril., 2006, 85, 1319-1340.
- L. Siciliano, P. Tarantino, F. Longobardi, V. Rago, D.C. Stefano, A. Carpino, J Androl., 2001, 22, 798-803.
- M. Murawski, J. Saczko, A. Marcinkowska, A. Chwikowska, M. Grybooe, T. Banaoe, Folia Histochemt. Cytobiol., 2007, 45, 123-126.
- H.P. Nissen, H.W. Kreysel, Klin Wochenschr., 1983, 61, 63-65.
- D. Sanocka, R. Miesel, P. Jedrzejczak, A.C. Chelmonska-Soyta, M. Kurpisz, Int. J. Androl., 1997, 20, 255-264.
- Y.Y. Hsieh, C. Chang, C.S. Lin, Int. J. Biol. Sci., 2006, 2, 23-29.
- Y.L. Huang, W.C. Tseng, S.Y. Cheng, T.H. Lin, Biol. Trace Elem. Res., 2000, 76, 207-215.