Research Article - Der Pharma Chemica ( 2021) Volume 13, Issue 3
Electrophilic Effects of Copper ions on kinetics of dissociation of Tris (1,10- Phenanthroline) iron (II) in Sodium dodecyl sulphate micellar medium
V TejeswaraRao1, K Balaji1, Satya guru TVSPV2 and S Raju3*2Vignan’s Institute of Information Technology (A), Duvvada, Visakhapatnam-530049, India
3Deapartment of Chemistry, Govt. Degree College, Chodavaram, Visakhapatnam, India
S Raju, Deapartment of Chemistry, Govt. Degree College, Chodavaram, Visakhapatnam, India, Email: tejavoonna@gmail.com
Received: 04-Mar-2021 Accepted Date: Mar 22, 2021 ; Published: 30-Mar-2021
Abstract
Many metal ions have been found to exert electrophilic effects on the dissociation of complexes and it has been proposed that a ternary intermediate is formed involving the complex and metal ion through the severing of one of the metal ligand bonds. To investigate this aspect, the dissociation of Fe (phen)32+ and effects of metal ion like Cu2+ in the presence of SDS micelles providing anionic micellar surface. These results confirm that the formation of such a ternary intermediate and marked micellar catalysis of the reaction.
Keywords
Spectrophotometric Determination, Cu2+, Tris (1,10- Phenanthroline) iron (II), Surfactant SDS
Introduction
The study of dissociation of tris (2,2-bipyridyl)iron(ll), [Fe(bipy)3]2+, and tris (1,10-Phenanthroline)iron(ll), [Fe(Phen)3]2+, attracted considerable attention of workers. Basolo et al [1] carried out a detailed kinetic study of dissociation of [Fe(bipy)3]2+, in the aqueous medium in the presence of acid. The dissociation of the complex is accelerated by H+ ion, the rate reaching a limiting value at [H+]=1.0 mol dm-3. Sriramam et al [2] also investigated the reaction in the aqueous acetic acid media including mineral acids and found a linear variation of rate with H+ ion concentration reaching a limiting value at [H+ ] =0.6 mol dm-3 at μ = 0.1 mol dm-3.
The solvent effect on the dissociation of [Fe(Phen)3]2+ and related complexes has been investigated in detail by Blandamer and Burgess and their co-workers [3-5]. A lot of work has been done on the kinetics of micelle-catalyzed dissociation or binding of iron(II) phenanthroline complex and its methyl substituted analogues, such as tris (3,4,7,8-tetramethyl-1,10-phenanthroline) iron(II) [6-14].
Micellar catalysis by surface-active agents or surfactants has become a subject of great significance because several aspects of this phenomenon exhibit features common with enzyme catalysis [15]. The presence of surface-active agent often brings about acceleration of reaction by 10-100 times and in a few cases 1000-10000 times [16-18].
Nucleophilic effects of anions on the complexes have been studied extensively [19-23] Subbarao et al. [24] reported that the dissociation of tris(2,2’-bipyridyl) iron(II), [Fe(bipy)3]2+, which is immeasurably slow in the aqueous medium in the absence of added acid, takes place with an appreciable rate reverse micellar medium and also stated that the added anions have accelerating effect on rate. Venkateswarlu and Krishna Rao investigated the Kinetics of the dissociation of tris(2,2-bipyridyl)iron(II) and tris(1,10-phenanthroline)iron(II) in the reverse micelles of Tween-85 in cyclohexane [25].
In addition to the nucleophilic effects of anions, electrophilic effects of metal ions on the complexes are also possible. Kopanica and Stara [26] reported the substitution reaction between hydrated Copper (II) and Cobalt(II) triethylenetetraminehexa acetate and the reverse reaction, in which binuclear intermediate has been proposed. Reports are also available in the literature [27-28] that metal ions exert Lewis acid effects on complexes weakening the coordinated bond making them amendable for nucleophilic attack.
From the studies of electrophilic effects of the metal ion on the dissociation of metal complexes, Subba Rao et al [29] found kinetic evidence for the ternary intermediate involving the dissociating complex and these metal ions.
It is interesting to investigate whether these reactions are further accelerated in the micellar media in which the micelle has been oppositely charged surface to that of the metal ion and the dissociating complex and whether the reaction proceeds through mechanism involving a ternary intermediate.
To investigate this aspect, the present author has chosen the dissociation of the cationic complex Fe(phen)32+and effects of Cu2+and Co2+metal ions in the presence of SDS micelles providing anionic micellar surface. These results are reported in this dissertation.
Preparation of Solutions
All the materials employed in this investigation were of analytical reagent grade. Solutions were prepared using water, distilled thrice in all-glass apparatus.
Tris (1,10-Phenanthroline) iron (ll)
BDH, AnalaR sample of 1,10-Phenanthroline was used for the preparation. The Complex was prepared by mixing stoichiometric amounts (molar ratio1:3) of ferrous ammonium sulphate (E. Merck) and 1,10-Phenanthroline in water.
Sodium dodecylsulphate (SDS)
Fluka sample of sodium dodecyl sulphate (SDS) has been used in the preparation of 0.1 mol dm-3 solution. The purity of the sample was tested by determining the critical micelle concentration (cmc) (8.0 X 10-3mol dm-3) conduct metrically.
PAN Indicator
Dissolve 50mg of the compound in 100ml of methanol or ethanol.
Copper sulphate
A known volume of copper solution is transferred into a conical flaskadd 10ml of acetate buffer (ammonium hydroxide- ammonium chloride, pH 10), 4-5 drops of PAN indicator, heat to boiling, and titrate with standard EDTA [30]. The color changes from blue-violet to yellowish green.
Kinetic measurements
The course of each of the reactions investigated has been followed by measuring the absorbance due to [Fe(phen)3]2+λmax = 510 nm (ε = 11,000 dm3 mol-1 cm-1) using Milton Roy spectronic 1201 uv-visible spectrophotometer with thermostatic arrangement.
Treatment of rate data
In all these reactions, plots of log At (absorbance attime‘t’) versus time have been found to be linear for atleast 98 % of the reaction. In all the experiments, the absorbance is due to the instantaneous concentration of[Fe(phen)3]2+ . The pseudo first order rate constants k1 have been obtained by multiplying slope of the plot with 2.303. All the kinetic runs have been carried out in triplicate and reproducible within ± 5%.
Product Analysis
The aqueous solutions of tris (1, 10-phenonthroline) iron (II) [Fe(phen)3]2+is intensely colouredand λmax = 510 nm (ε = 11,000 dm3 mol-1 cm-1)}. In the dissociation of [Fe(phen)3]2+ the products of dissociation are Fe2+(aq) and 1,10-phenanthroline.The dissociation has been established by the decrease in absorbance of the complex at λmax = 510 nm with time, where the products of dissociation Fe2+(aq) and phenhave no absorbance. To the product, ammonium thiocyanate was added and no red colour is developed indicating the absence of Fe(III), the products being Fe2+(aq) and phen, where phen = 1, 10-phenanthroline.In accord with this the UV- visible spectra of the reaction mixtures at the end of the reaction overlap with those of 1, 10-phenanthroline at identical concentrations.
Determination of the CMC of Sodium Dodecyl Sulphate Under the Experimental Conditions Employed
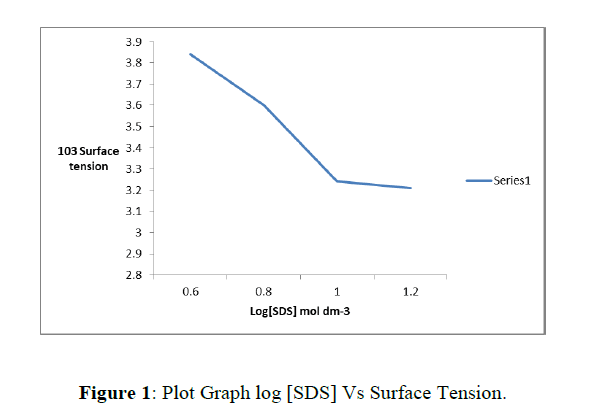
The critical micelle concentration of SDS has a value of 8.0 X10-3 mol dm-3 in the absence of any added ions [31], but the value decreases as the ionic strength of the medium increases. The author has determined CMC at μ = 0.1 mol dm-3with respect to sodium per chlorate and in the presence of [Fe(phen)3]2+= 4.0 X10-5mol dm-3 by determining the surface tension of the mixture at different concentrations of SDS using stalagmometre. The value of surface tension is plotted against logarithms of SDS concentration. The decrease in surface tension with increase in [SDS] is very pronounced at SDS, the decrease in surface tension is much less pronounced. The plot has been found to yield two straight lines intersecting at [SDS] = 1.0 X10-3mol dm-3 and hence, the CMC under experimental conditions has been taken to be 1.0 X10-3mol dm-3 (Figure 1). This result is in agreement with the CMC reported under similar experimental conditions [32-33] (Table 1).
| 10-3[SDS]o mol dm-3 |
103( Surface Tension) Jm-2 |
|---|---|
| 0 | 6.48 |
| 0.4 | 3.97 |
| 0.6 | 3.71 |
| 0.8 | 3.49 |
| 1 | 3.29 |
| 1.2 | 3.27 |
| 1.5 | 3.24 |
| 2 | 3.19 |
| 2.5 | 3.11 |
| 3 | 3.11 |
| 3.5 | 3.11 |
| 4 | 3.11 |
[Fe(Phen)3+2]=4.0 x 10-5mol dm-3 Temp.=25 ± 0.1°C
Table 1: Determination of CMC.
Effect of variation of [Cu2+] ion on rate in aqueous medium
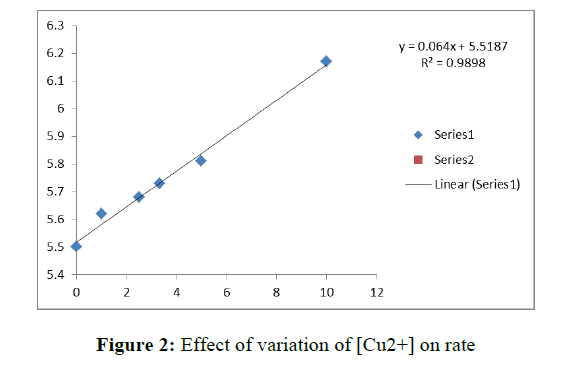
To investigate the effect of concentration of the Cu+2 on the rate of dissociation of Fe(Phen)32+ in the aqueous medium, kinetic runs were carried out in the presence of different concentrations of each of the metal ion, at constant ionic strength. The pseudo first order rate constants k’ are presented in Table 2. These results show that k’ increases with increase in the metal ion concentration and shows limiting behavior at higher metal ion concentration. The plots of 1/k’ Vs 1/ [metal ion] are linear with positive intercepts (Figure 2).
| 103 [Cu2+] (mol dm-3) |
104 .k’ (Sec-1) |
|---|---|
| 1 | 1.62 |
| 2 | 1.71 |
| 3 | 1.74 |
| 4 | 1.76 |
| 10 | 1.78 |
[Fe(Phen)32+] = 4.0 X 10-5 mol dm-3 Temp = 25.0 ± 0.1 μ = 0.01 mol dm-3
Table 2: Effect of variation of [Cu2+] on rate.
Effect of variation of metal ion concentration on rate in presence of SDS micelles
To investigate the effect of concentration of each of the metal ion Cu2+ on the rate of dissociation of Fe(Phen)32+ in the anionic micellar medium, kinetic runs were carried out in the presence of different concentrations of each of the metal ion, at constant ionic strength and at constant [SDS].
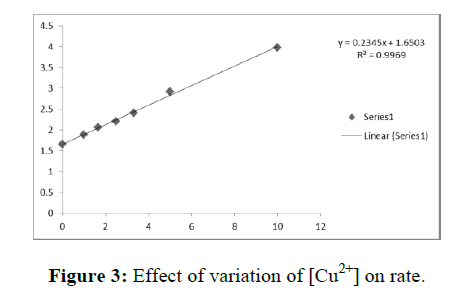
The pseudo first order rate constants k’ are presented in Table 3. These results show that k’ increases with increase in the metal ion concentration and shows limiting behavior at higher metal ion concentration. The plots of 1/k’ Vs 1/ [metal ion] are linear with positive intercepts (Figure 3). This shows that kinetic features of the reactions are similar in aqueous and micellar media.
| 104 [Cu+2] (mol dm-3) |
104 .k’ (Sec-1) |
|---|---|
| 1 | 2.52 |
| 2 | 3.43 |
| 3 | 4.167 |
| 4 | 4.54 |
| 6 | 4.87 |
| 10 | 5.32 |
[Fe(Phen)32+] = 4.0 X 10-5mol dm-3[SDS] = 2.0 X 10-3mol dm-3
Table 3: Effect of variation of [Cu2+] on rate
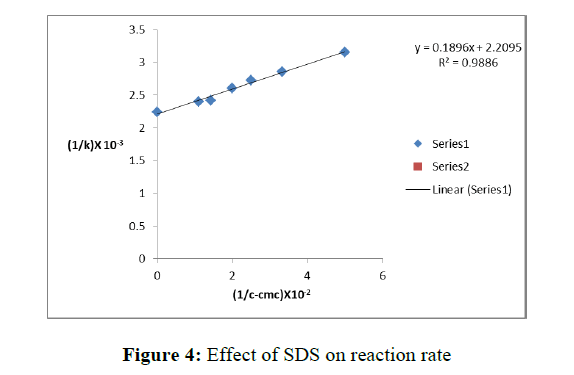
To investigate the effect of [SDS] on the rate of the reaction, kinetic runs were carried out at constant ionic strength by varying [SDS] but keeping the concentrations of Cu2+or Co2+constant. The pseudo first order rate constants k’ are presented in Table 4.
| 103 [SDS] mol dm-3 | 104 .k’ (Sec-1) |
|---|---|
| 3 | 3.17 |
| 4 | 3.5 |
| 5 | 3.68 |
| 6 | 3.84 |
| 8 | 4.13 |
| 10 | 4.17 |
[Fe(Phen)32+] = 4.0 X 10-5mol dm-3 [SDS] = 2.0 X 10-3mol dm-3
[Cu2+]=1.0 X 10-4mol dm-3 Temp = 30°c μ = 0.01 mol dm-3
Table 4: Effect of SDS on reaction rate.
The results in table 4 show that the reaction rate appreciably increases in the micellar medium. In presence of anionic SDS micelles, the rate of reaction increases by 8 times in case of Copper ions. Further, rate constants show a tendency towards limiting behavior at higher [SDS], but no maximum is observed (Figure 4).
Results and Discussions
Kinetics of dissociation of [Fe(Phen)3]2+ in aqueous phase
The salient kinetic features in the aqueous phase are: The reaction obeys first order kinetics with reference to Fe(Phen)32+ but fractional order kinetics with respect to the metal ion (Cu2+or Co2+). The plot of 1/kobsVs 1/[M]T is linear with a positive intercept.
The results shown that in the presence of the metal ions, the pseudo first order rate constants show tendency of limiting behavior at high [metal ion], indicating the formation of an intermediate 1:1 add between metal ion and [Fe(phen)3]2+, probably one of the coordinate bonds of the complex, [Fe(phen)3]2+ , is severed the freed nitrogen than forming a coordinate bond with the metal ion concomitantly. The metal ion in this ternary intermediate may influence further severing of the iron (II) – 1, 10-phenanthrolin bonds through its electophilic effect.
To study the stability of tris1,10-phenanthroline Iron(II) at pH = 6.8 (where the present investigations have been conducted) the absorbance has been determined at various times, and the author found that there is no change in absorbance evenafter6 hours due to dissociation of complex. Hence this component needs not to be considered in the analysis of rate data.
The k’ versus [M] plots show limiting behavior at higher metal ion concentration and the plots of 1/k1Vs 1/[Metal ion] are linear with the positive slopes and intercepts.
Thus the present author has found that in addition to Ni2+, Zn2+, other ions like Co2+,Cu2+also accelerates the dissociation of Fe(phen)32+ and the kinetic results prove the formation of ternary intermediate involving Fe(phen)32+-M2+.The metal ion (Cu2+or Co2+) in the ternary intermediate exerting the electrophilic effect and accelerating the decomposition of Fe(phen)32+. The authors results further established that the negative SDS micelles further accelerates the reaction and the mechanism and kinetic pattern with micellar phase are similar to those in aqueous media.
References
- Basolo F, Hayes JC and Neumann HM. J Am Chem Soc. 1954, 76: p. 3807.
- Sriramam K, Sreelakshmi J, Ramadevi L et al., Int J Chem Kin. 1992, 24: p. 11.
- Blandamer MJ, Burgess J, Cope SD et al., T Trans Met Chem. 1984, 9: p. 347.
- Blandamer MJ, Burgess J and Clark B. J Chem Soc Chem Com. 1983, p. 659.
- Burgess J. J Chem Soc(A). 1968, p.1085.
- Tachiyashiki S and Yamatera H. Polyhedron. 1983, 2: p. 9.
- Lee TS, Kolthoff IM, Leusing DL. J Am Chem Soc. 1948, 70: p. 3596.
- Brandt WW and Gullstrum DK. J Am Chem Soc. 1952, 74: p. 3532.
- Magerum DW. J Am Chem Soc. 1957, 79: p. 2728.
- Burrows HD, Ige JW, Umoh SA. J Chem Soc Faraday Trans I. 1982, 78: p. 9447.
- Ige J and Soriyan OO. J Chem Soc Faraday Trans I. 1986, 82: p. 2011.
- Ige J, Lambi JN and Soriyan OO. J Chem Soc Faraday Trans I. 1988, 1: p. 84.
- Ige J and Soriyan O. React Kinet Catal Lett. 1993, 49: p. 419.
- Ige J and Soriyan O. React Kinet Catal Lett. 1993, 49: p.427.
- Berezin IV, Martinek K and Yatisimirskii AK. Russ Chem Rev. 1973, 42: p. 787.
- Cordes EH and Dunlop RB. Acct Chem Res. 1969, 2: p. 329.
- Fendler EJ and Fendler JH. Academic press, Newyork. 1975.
- Yatsmirskii AK, Martinek K and Berezin IV. Tatrahedron. 1971, 27: p. 2855.
- Tachiyashiki S and Amatara HY. Polyhedron. 1983, 2: p. 9.
- Venkateswarlu G, Syamala P, Subbarao PV et al., J Indian Chem Soc. 2003, 86: p. 90.
- Syamala P, Subbarao PV and Ramakrishna K. Ind J Chem. 2000, 39: p. 643-645.
- Deepa Sarkar K, Subbarao PV, Gousia Begum et al., J Colloid Interface Sci. 2005, 288: p. 591-596.
- Deepa Sarkar, Gousia Begum, Ramakrishna K et al., J Surface Sci Technol. 2004, 20: p. 9-18.
- Venkateswarlu G, Syamala P, Subbarao PV et al., Ind J Chem. 2002, 41: p. 1410-1414
- Venkateswarlu G and Krishna Rao GSR. J Indian Chem Soc. 2009, 86: p. 826-831.
- Kopanika M and Stara V. Chem Comm. 1972, 37: p. 80.
- Das, Radha R, Bhat TR et al., J Inorg Nucl Chem. 1968, 30: p. 1691.
- Huges MN. The Inorganic chemistry of Biological process, 2ndEdn. (Wiley), 1988.p. 80.
- Phanikrishna PS, M.Phil Dissertation, Andhra University, 2010.
- Kolthoff IM, 4th Edition, Published by The MacMillan Company(London), p. 808.
- Bunton CA and SavelliG, Advances in Physical organic Chemistry, 1986. 22: p. 216.
- Bunton CA and Wolfe B. J Am Chem Sco. 1973, 95: p. 3742.
- Pramauro E, Pelizzetti E, Diekmann S et al., Inorg Chem. 1982, 21: p. 2432.