Research Article - Der Pharma Chemica ( 2024) Volume 16, Issue 4
Efficient Synthesis of 3-(phenyl sulfonyl)-2H-Chromen-2-one
R. V. Kupwade*R. V. Kupwade, Department of Pharmaceutics, Kasturbai Walchand College of Arts and Science, Sangli, India, Email: rv_kupwade@yahoo.co.in
Received: 16-Jul-2024, Manuscript No. DPC-24-145255; Editor assigned: 19-Jul-2024, Pre QC No. DPC-24-145255 (PQ); Reviewed: 26-Jul-2024, QC No. DPC-24-145255; Revised: 01-Aug-2024, Manuscript No. DPC-24-145255 (R); Published: 29-Aug-2024, DOI: 10.4172/0975-413X.16.4.425-430
Abstract
Herein enviro-economic protocol for the synthesis of 3-phenylsulfonyl coumarins using THAM as a commercially available catalyst has been reported. Decent yields and easy isolation of the products avoiding conventional isolation as well as purification methods are the noteworthy merits of the developed protocol. The most fascinating and significant finding regarding driving force in the synthesis of phenylsulfonyl coumarins through Knoevenagel condensation reaction has also been disclosed.
Keywords
Coumarin; Knoevenagel condensation; Cyclization; Phenyl sulfonyl acetonitrile; THAM
Introduction
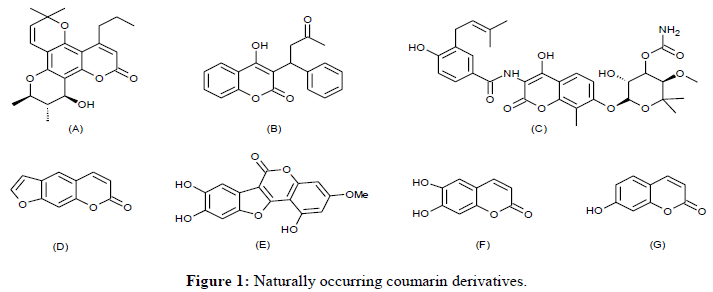
Coumarin constitutes a core unit of diverse natural products that exhibit a range of biological activities [1]. Coumarin was first isolated in 1820 from Tonka beans [2]. Large number of coumarin derivatives are distributed in high concentration in Tonka bean, lavender, licorice, woodruff, apricots, cherries, strawberries, sweet clover, cassia leaf oil, cinnamon bark oil and bison grass having vanilla like flavor (Figure 1).
Calanolide-A (Figure 1A) is a natural coumarin shows non-nucleoside reverse transcriptase inhibitor (NNRTI) and is known to exhibit anti- HIV activity [3,4]. Warfarin (Figure 1B) is known to exhibit anticoagulant property and it is used to prevent clotting of blood in the veins and heart [5]. Novobiocin (Figure 1C), a 3-benzamidocoumarin derivative which possesses strong antibiotic activity by inhibiting DNA gyrase and has potential as an anticancer agent [6]. Psoralens (Figure 1D) are mainly used in the treatment of psoriasis [7]. Wedelolactone (Figure 1E) is another naturally occurring furan ring fused coumarin and is used as a venomous snake-bite antidote [8]. A natural coumarin analogue, Esculetin, 6,7-dihydroxycoumarin (Figure 1F) has antioxidant, antiproliferative and anti-inflammatory activities [9] while Umbelliferone, 7- hydroxycoumarin (Figure 1G) is encountered in several plants with notable antimycobacterial [10] and anticancer [11] activities.
Coumarins in general have effective healing action against edema and are able to subsidize swelling. Therefore, they are also used in the treatment of lymph edema, elephantiasis and other high protein edema conditions [12]. Several synthetic compounds which enclose coumarin moiety are well known for their odour, availability and stability. They are commonly used in perfume, soaps, detergents, [13] optical brightening agents [14] and in the synthesis of insecticides. Furthermore, the pharmacological, biochemical and therapeutic properties of simple coumarins can be altered by the incorporation of particular group as a fused component or through their structural modifications.
Variety of reactions like Wittig reaction, Perkin reaction, Pechmann reaction, Michael reaction, Claisen rearrangement, Reformatsky reaction are known for coumarin synthesis. However, the simplest route towards the synthesis of coumarins involves a base catalyzed reaction of ohydroxy benzaldehyde with active methylene compounds like diethyl malonate, ethyl acetoacetate, ethyl cyanoacetate, etc. The reaction follows Knoevenagel condensation intramolecular cyclization path and furnishes 3-acetyl, 3-carbalkoxy or 3-cyano coumarin respectively [15,16]. Phenylsulfonyl acetonitrile (pKa=12) is also be the choice as active methylene compound. The importance of phenylsulfonyl acetonitrile is associated with the advantages that the cyano group can be easily transformed into other functionalities while sulfonyl group being a good leaving group, can be removed easily by the reactions like hydrolysis, reduction or substitution reaction [17]. However, there are only few reports on the choice of phenyl sulfonyl acetonitrile as an active methylene compound in the synthesis of coumarins [18]. In this report synthesis 3-phenylsulfonyl coumarins [3-(phenyl sulfonyl)-2H-chromene-2-one] has been explored.
Materials and Methods
Experimental
General: Simple and substituted salicylaldehyde were bought from commercial sources and were used as received. Phenyl sulfonyl acetonitrile was synthesized by the sulfonylation of cyanomethyl phenyl sulfide (Phenyl sulfanyl acetonitrile). Melting points were recorded using Kumar melting point apparatus. 1H and 13C NMR spectra were recorded using Bruker Avance-II (300 MHz) spectrometer.
Representative procedure for sequential one pot, two step synthesis of 3-phenylsulfonyl coumarins: To a well stirred solution of salicylaldehyde (1 mmol) and phenylsulfonyl acetonitrile (1 mmol) in ethanol (95%, 5 mL) was added THAM (20 mol %). Stirring was continued and the reaction was monitored by TLC. Upon completion of the reaction (TLC), 2N HCl (5 ml) was added to resultant iminochromene and the reaction mixture was refluxed for two to three hrs more. After complete consumption of iminochromene evidenced by TLC, reaction mixture was cooled to room temperature. The final product 3-phenylsulfonyl coumarin was obtained as fine suspended solid. It was simply filtered, washed with water, dried, washed again with hexane-chloroform mixture (3:1, v/v) and dried. The resultant product was found to be pure and did not require any further purification.
From retro synthetic analysis, synthesis of 3-phenylsulfonyl coumarin can be achieved using salicylaldehyde and phenylsulfonyl acetonitrile as precursor. From mechanistic point of view, the success of the synthesis of 3-phenylsulfonyl coumarins depends upon yield of Knoevenagel condensation product. Therefore, initially protocols available for Knoevenagel condensation reaction between aromatic aldehydes and phenylsulfonyl acetonitrile were investigated. Very few reports on this condensation reaction were found. Most of the reported protocols are operable at elevated temperature while a few others suffer from the shortcomings such as poor yields, workup process, etc. Therefore exploration of a suitable base catalyst for Knoevenagel condensation of aldehydes with phenylsulfonyl acetonitrile at ambient temperature is desirable.
Results and Discussion
In this context, anisaldehyde and phenylsulfonyl acetonitrile were selected as the model substrates and few model reactions were carried out with equimolar quantities of anisaldehyde, 1 and phenylsulfonyl acetonitrile, 2 (2 mmol, each) were stirred together in ethanol medium the presence of various basic catalysts (Table 1).
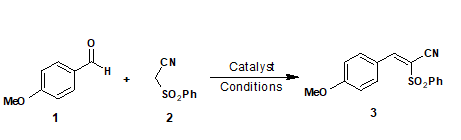 Anisaldehyde Phenylsulfonyl acetonitrile |
|||
|---|---|---|---|
| Entry | Catalyst (mol %) | Time (h) | Yield (%)b |
| 1 | K3PO4 (20) | 24 | 50 |
| 2 | K2CO3 (20) | 24 | 30 |
| 3 | MgO (0.5 g) | 24 | 20 |
| 4 | Et3N (20) | 3 | 80 |
| 5 | Pyrrolidine (20) | 75d | 80 |
| 6 | Piperidine (20) | 2 | 93 |
| 7 | DABCO (20) | 60c | 78 |
| 8 | DBU (20) | 2.5 | 90 |
| 9 | Et2NH (20) | 5 | 75 |
| 10 | THAM (20)e | 8 | NR |
Note: a: Reaction conditions: Anisaldehyde and phenylsulfonyl acetonitrile (2 mmol, each), catalyst, ethanol (5 mL), RT; b: Isolated product; c: Time in minutes; d: Reflux; e: Tris-hydroxymethylaminomethane
Table 1: Screening of catalyst for Knoevenagel condensation between anisaldehyde and phenylsulfonyl acetonitrilea.
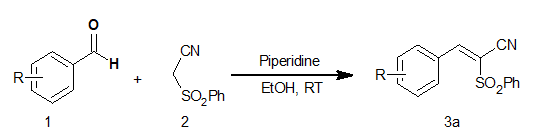
Based upon these results (Table 1), piperidine was observed to be the best suitable catalyst. It is worthy to note that, THAM that serves as a very weak base failed to furnish the desired product. Piperidine-ethanol was confirmed as the best suited catalyst-solvent combination for Knoevenagel condensation between anisaldehyde and phenylsulfonyl acetonitrile. Upon completion of piperidine catalyzed model reaction, resultant product was isolated and characterized by physical (melting point) as well as chemical methods. Next, to establish the scope as well as generality of the protocol, aromatic aldehydes carrying electron-donating as well as electron-withdrawing groups were employed to undergo condensation reaction with phenylsulfonyl acetonitrile. In all the cases, desired condensation product, 3b-k, were obtained in excellent yield and purity (Table 2).
 Aldehydes Phenylsulfonyl acetonitrile |
|||||
|---|---|---|---|---|---|
| Product | Aldehyde (1) | Time (h) | Yield (%)b | M. P. °C | |
| Obs. | Lit.Ref. | ||||
| 3a | 4 - Methoxybenzaldehyde | 1.5 | 94 | 110-112 | 113 - 11518c |
| 3b | Benzaldehyde | 1.5 | 88 | 135-139 | 130-13218b |
| 3c | 4-Methylbenzaldehyde | 1.5 | 92 | 148-151 | 144-14618d |
| 3d | 4-Chlorobenzaldehyde | 1.5 | 80 | 156-158 | 152-15418d |
| 3e | 3,4-Dimethoxybenzaldehyde | 1.5 | 95 | 152-155 | --- |
| 3f | 4-Cyanobenzaldehyde | 1 | 82 | 189-192 | --- |
| 3g | 4-Bromobenzaldehyde | 1.5 | 89 | 146-148 | --- |
| 3h | 3-Nitrobenzaldehyde | 1 | 85 | 150-152 | 150-15218c |
| 3i | Piperonal | 1 | 99 | 172-175 | 170-17218c |
| 3j | Thiophene-2-carbaldehyde | 1.5 | 87 | 114-115 | --- |
| 3k | 4-Hydroxybenzaldehyde | 1.5 | 89 | 218-221 | --- |
Note: a: Reaction conditions: Aldehyde and phenylsulfonyl acetonitrile (2 mmol, each), catalyst (20 mol %), ethanol (5 mL), RT; b: Isolated product
Table 2: Piperidine catalyzed Knoevenagel condensation between aldehydes,1, and phenylsulfonyl acetonitrile, 2a.
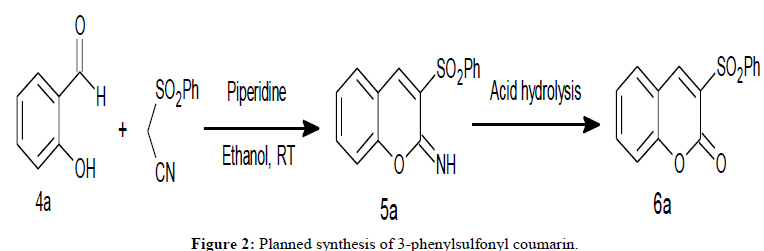
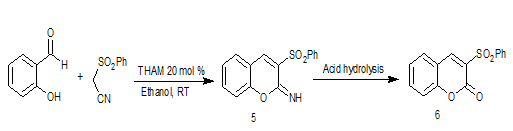
Confirmation of structures of all the Knoevenagel condensation products was done by comparison of their melting points with those reported earlier as well as by spectroscopic methods. After establishing the reaction conditions for Knoevenagel condensation between aldehydes and phenylsulfonyl acetonitrile, the protocol was extended towards the synthesis of 3-phenylsulfonyl coumarin, 6a. It was surmised that, piperidine catalyzed reaction of salicylaldehyde with phenylsulfonyl acetonitrile would furnish intermediate imino coumarin, 5a and the same upon it’s acid hydrolysis would furnish 3-phenylsulfonyl coumarin, 6a (Figure 2).
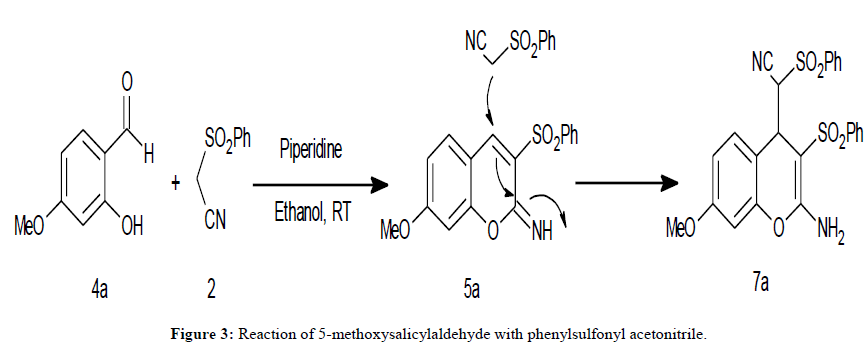
In an exemplar reaction, to a well stirred solution of 2-hydroxy, 4-methoxybenzaldehyde, 4a and phenylsulfonyl acetonitrile (2 mmol, each) in ethanol (5 mL) was added piperidine (20 mol %) as the catalyst. Stirring was continued at ambient temperature and the reaction was monitored by TLC. Upon completion of the reaction, the reaction mixture was diluted with water and the resultant solid product was isolated by filtration. TLC examination indicated the presence of two products, one, in major product and another in minor amount ( 80:20, TLC).
These products were separated by column chromatography. On the basis of physical as well as spectral methods the major product was identified to be 7-methoxy-3-(phenylsulfonyl)-2H-chromene-2-imine, 5a. On the other hand, the product obtained in minor amount was identified to be 2-amino-3-phenylsulphonyl-6-methoxy-4H-chromen-4-yl–benzenesulphonyl acetonitrile, 7a. The product, 7a is reported earlier to result by base catalyzed reaction of 2-hydroxy-4-methoxy-benzaldehyde with two equivalents phenylsulfonyl acetonitrile following Knoevenagel condensation-cyclization-michael addition cascade pathway. In this model reaction we have noticed the formation of 7a even with use of only one equivalent of phenylsulfonyl acetonitrile (Figure 3).
After the separation of these two products viz. 5a and 7a it was quite easy to convert 7-methoxy-3-(phenylsulfonyl)-2H-chromene-2-imine, 5a, to the desired product viz. 7-methoxy-3-phenylsulfonyl coumarin, 6a, by simple acid hydrolysis. However, exclusive formation of 5a was not achieved which will avail the possibility of sequential, one-pot synthesis of 3-phenylsulfonyl coumarins, 6a.
Hence, once again the optimization of reaction conditions for the reaction between 2-hydroxy-4-methoxy benzaldehyde and phenylsulfonyl acetonitrile was done to avoid formation of side product 7a. In this context, very weak base was used as the catalyst, which will promote Knoevenagel condensation between salicylaldehyde and phenylsulfonyl acetonitrile but will not promote subsequent michael addition reaction.
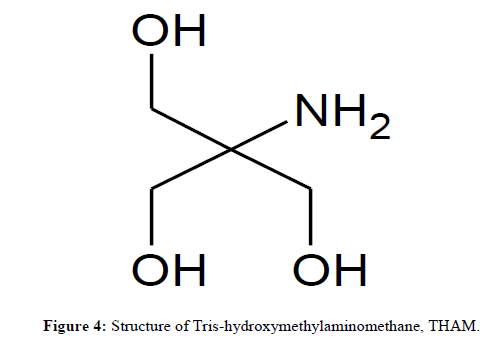
It is well known that, Tris-hydroxymethylaminomethane, THAM (Figure 4) is a biodegradable, non-corrosive, physiologically inert and thermally stable compound available commercially at extremely low cost. Customarily, it is used as an excipient in pharmaceutical preparations and in the analysis of pharmaceuticals. In water or water-ethanol medium, it generates basic reaction medium (pH 7.5 to 8.5). The catalyst has earlier been explored for the synthesis of tetrahydrobenzo[b]pyrans as well as pyran-annulated heterocycles. This clued up to test the efficacy of THAM in the synthesis of 3-(phenylsulfonyl)-2H-chromene-2-imine, 5.
In a model reaction, to an equimolar (1 mmol, each) and well stirred solution of 2-hydroxy-4-methoxy benzaldehyde, 4a and phenylsulfonyl acetonitrile in ethanol (5 ml) was added THAM (20 mol %). Stirring was continued at ambient temperature till completion of the reaction (monitored by TLC). The product was isolated by dilution of the reaction mixture with water followed by filtration. Resultant product in almost quantitative yield was noticed to be single compound and that too the desired product, 5a (TLC, NMR comparison).
In light of earlier results about the failure of THAM to promote Knoevenagel condensation between anisaldehyde and phenylsulfonyl acetonitrile (Table 3) and the success in exclusive formation of 5a as the product of reaction between salicylaldehyde and phenylsulfonyl acetonitrile following Knoevenagel condensation intramolecular cyclization path was a big surprise. And, this led to conclude that, during the formation of 5a, intramolecular cyclization is the real driving force for the Knoevenagel condensation between salicylaldehyde and phenylsulfonyl acetonitrile.
After getting 7-methoxy-3-(phenyl sulfonyl)-2H-chromene-2-imine, 5a, as an exclusive product it is easily converted into to the desired product, 3-phenylsulfonyl coumarin, 6a, by conventional acid hydrolysis. Accordingly, to a stirred suspension of 5a in water (2 ml) was added 2N HCl (5 ml) and the reaction mixture was refluxed till hydrolysis was complete (TLC). Upon cooling the reaction mixture, resultant solid product was filtered.
|
|||||
|---|---|---|---|---|---|
| Entry | Product | Time (h) | Yield (%)b | M. P. 0C | |
| Obs. | Lit. Ref. | ||||
| 6a |
|
8 | 97 | 217-219 | --------- |
| 6b | 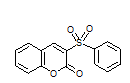
|
7.5 | 95 | 215-216 | 214-215 17a |
| 6c |
|
8 | 96 | 208-210 | --------- |
| 6d |
|
7 | 96 | 260-261 | --------- |
| 6e | 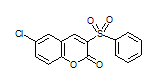
|
7.5 | 90 | 244-246 | 242-244 17a |
| 6f |
|
7.5 | 89 | 252-253 | --------- |
| 6g |
|
7 | 96 | 245-246 | --------- |
| 6h | 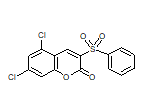 |
7 | 96 | 235-236 | --------- |
| 6i |
|
7 | 96 | 257-258 | --------- |
| 6j |
|
8.5 | 78 | 273-275 | --------- |
Note: a: Reaction conditions: Salicylaldehyde and phenylsulfonyl acetonitrile (2 mmol, each), THAM (20 mol %), ethanol (5 mL), RT; add 2N HCl (5 ml) reflux
Table 3: Sequential one-pot, two step synthesis of 3-phenylsulfonyl coumarinsa.
TLC examination indicated that, resultant product was pure and did not require any further purification. On the basis of comparison of its melting point with that reported earlier it was identified to be the desired product 7-Methoxy-3-(phenylsulfonyl)-2H-chromene-2-one, 6a. The result obtained during the synthesis of 6a following sequential, one-pot process was then substantiated by performing the reaction of variety of substituted salicylaldehydes and phenylsulfonyl acetonitrile. In all cases, desired 3-phenyl sulfonyl coumarins were obtained in excellent yield as well as purity. The results are summarized in Table 3. The structures of all the products were confirmed by spectroscopic methods.
Conclusion
A plethora of methods have earlier been reported for the synthesis of coumarins. However, reports on the synthesis of 3-phenyl sulfonyl coumarins are scanty. This observation impelled to uncover the reason behind not selecting phenylsulfonyl acetonitrile in the synthesis of coumarins. In this context the development of an enviro economic protocol for the synthesis of 3-phenylsulfonyl coumarins using THAM as a commercially available catalyst has been successfully reported. The wide scope, ambient reaction conditions and avoidance of chromatographic purification are the noteworthy features of the developed protocol. Perhaps this is the simplest and eco friendliest protocol designed for the synthesis of targeted compounds. The most important outcome of this work is the observation that, rather than Knoevenagel condensation, intramolecular cyclization is the real driving force in the formation of 3-phenylsulfonyl coumarin.
References
- O’Kennedy R, Thomas RD. Coumarins: Biology, Applications, and Mode of Action. Wiley & Sons: Chichester. 1997: p. 360.
- Xu ZQ, Flavin MT, Jenta TR. Curr Opin Drug Discov Devel. 2000; 3(2): p. 155-166.
[Google Scholar] [PubMed]
- Hirsh J, Fuster V, Ansell J, et al. J Am College Cardio. 2003; 41(9): p.1633.
- Raad II, Hachem RY, Abi-Said D, et al. Cancer. 1998; 82: p. 403.
[Crossref]
- Diogo LC, Fernandes RS, Marcussi S, et al. Basic Clin Pharmacol Toxicol. 2009; 104(4): p. 293-299.
[Crossref] [Google Scholar] [PubMed]
- Witaicenis A, Seito LN, Di Stasi LC. Chem Biol Interact. 2010, 186: p. 211-8.
- Ramalingam R, Vaiyapuri M. J Acute Med. 2013; 3(3): p. 73-82.
- Casley-Smith JR, Casley-Smith JR. John Wiley Sons, Inc, New York, NY. 1997; p.348.
- Gottlieb OR, Herrmann K, Murray RDH, et al. Progress Chem Org Nat Prod, Springer-Verlag Wein. 1978: p. 200.
- Zabradnik M. The production and application of fluorescent brightening agents. Wiley, New York. 1992.
- Little RD, Myong SO. Tetrahedron Lett. 1980; 21: p. 3339.
- Merchant JR, Shah P. J Heterocyclic Chem. 1981; 18: p. 441.
- Climent MJ, Corma A, Guil-Lopez R et al. J Catal Lett. 1999; 59: p. 33.
- Kulkarni MA, Pandit KS, Desai UV, et al. Comptes Rendus Chimie. 2013; 16(8): p. 689-695.
- Taha M, Lee M. J Phys Chem Chem Phys. 2010; 12: 12840.
[Crossref]
- Kramancheva I, Dobrev I, Brakalov L, et al. Anal Lett. 1997, 30(12): p 2235–2249.
[Crossref]
- Pandit KS, Chavan PV, Desai UV, et al. New J Chem. 2015; 39(6): p. 4452-4463.
- Kupwade RV, Khot SS, Lad UP, et al. Res Chem Intermed. 2017; 43: p. 6875-6888.