Research Article - Der Pharma Chemica ( 2018) Volume 10, Issue 1
Adrenomedullin: A Novel Vasoregulatory Peptide Hormone Enhances Invasion of Trophoblast on Uterine Endometrium-The First Trimester Human Trophoblast ACH-3P Cell Line on Early Pregnancy
Kanchi Ravi Padma, Penchalaneni Josthna*
Department of Biotechnology, Sri Padmavati Mahila Visvavidyalayam (Women’s University), Tirupati, Andhra Pradesh-517502, India
- Corresponding Author:
- Penchalaneni Josthna
Department of Biotechnology, Sri Padmavati Mahila Visvavidyalayam (Women’s University), Tirupati, Andhra Pradesh-517502, India
Abstract
Calcitonin Gene-related Peptide (CGRP), amylin and Adrenomedullin (ADM) belong to a distinctive group of calcitonin (CGRP-α)/CGRP-β) family peptides that have related biological outcome owing to their structure and cross-reactivity between receptors. We recently reported that the level of immune reactive ADM is significantly up regulated in pregnant rats during early implantation period. Furthermore, infusion of ADM antagonist in pregnant rats causes uteroimplantation growth restriction. The purpose of this study was to analyze the expression and possible role of ADM in ACH-3P first trimester cell lines. ADM is localized in syncytiotrophoblast, cytotrophoblast and endothelial cells throughout human pregnancy. This study also established that ADM enhances the invasion and migration of first-trimesterACH-3P cells. The present work mainly concerned with the cell line analysis to investigate cytotoxicity extent determination by 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) assay. The cell lines initially subjected to the MTT assay to determine the cytotoxicity effect of ADM22-52. DNA fragmentation is exposed by the presence of a multitude of DNA strand breaks in TUNEL assay by flow cytometric method. As a whole, the results from this study suggested that ADM have animperativerole in the physiology of human pregnancy via regulation of trophoblast invasion and migration.
Keywords
Trophoblast, MTT, Placenta, Pregnancy, Syncytiotrophoblast
Introduction
Trophoblast migration and invasion play fundamental roles in the process of embryo implantation and placentation. Any commotion of embryo implantation and placentation can lead to many pregnancy complications, such as placenta increta, recurrent spontaneous abortion, preeclampsia and premature delivery. Moreover the trophoblast invasion into the uterine endometrium is important for placentation [1,2]. As soon as implanted, the cytotrophoblast which are first formed differentiate into the villous cytotrophoblast and the Extra Villous Cytotrophoblast (EVCT). The earlier formed cytotrophoblast combine to form the multinucleated syncytiotrophoblast conscientious for fetomaternal exchange and production of hormones. The syncytiotrophoblast form migratory cell columns that infest the endometrium [3]. EVCT invasion peaks at early pregnancy and demur thereafter [4]. The placenta is an exclusive, temporary organ developed exclusively for the fetus. A fetus continues to exist with a wide diameter, low-resistance vascular system for adequate blood flow. The cytotrophoblast cell performs a significant role in establishing maternal-fetal circulation for the development of the fetus. The cytotrophoblast cells are derived from the trophectoderm, the outermost epithelial cell layer of the blastocyst. The invasion of EVCT engrosses cellular proliferation, entrenchment of the cells and degradation of extracellular matrix, and migration through connective tissue [5]. On the contrary to tumorinvasion, EVCT invasion is spatially and temporally regulated, and confined to the inner third of the myometrium [5]. Although intensive research on the mechanisms controlling human EVCT invasion are feebly understood, and are believed to be mediated by delicate balance between stimulatory and inhibitory factors of both trophoblastic and uterine origins [6]. Abnormalities in these factors lead to ineffective invasion as in miscarriage, preeclampsia, and intrauterine growth restriction, or excessive invasion as in choriocarcinoma [7-9].
In humans the expression of Adrenomedullin (ADM) was identified in trophoblast and decidua of placenta during the first trimester of pregnancy and in vitro studies of human EVCT derived cell lines demonstrates the role of endogenous ADM in invasiveness, cell migration, proliferation and in the invasion of EVCT modulatory activity of ADM on human EVCTs [10,11]. ADM was also involved in rodent embryogenesis and its expressions were also observed in the heart, neural, and skeletal-forming tissues as well as in the main embryonic internal organs [12]. All these observations implicate the involvement of ADM during placentation and fetal growth in human pregnancy; however, it is unclear whether this involvement is critical. Taking into account in our previous work using ADM antagonist we demonstrated the importance of endogenous ADM during early gestation and its role in regulation of apoptosis in fetoplacental development [13]. In accordance to our previous reports it came to known that endogenous ADM taking involvement in reproductive functions and in development of fetus during pregnancy and moreover various studies have shown the endogenous ADM expressions and its plasma levels throughout the pregnancy but there was no particular in vitro studies to prove ADM function as significant hormone for implantation and the placentation. On these grounds, the present study was designed to evaluate the involvement of endogenous ADM during implantation and in control of blastocyst proliferation through apoptosis.
Several trophoblast cell lines have been recognized and portrayed. All of them have diverse features and each of them may bear a resemblance to distinct trophoblast subpopulation. The ACH-3P first trimester trophoblast cell line has high proliferation activity which is significant for our study. They are imperative for investigations on the specific trophoblast subpopulations. However, there are limited studies carried out on ACH- 3P cell line, since they are of limited value for studies into DNA fragmentation and Tumor Necrosis Factor Alpha (TNF-α) signaling pathway that interplay between subpopulations of trophoblast.
Materials and Methods
Cell culture
ACH-3P (American Type Culture Collection) is a immortalized cell line from human [14] and is frequently used as model for the study of EVCT invasion [15,16]. The immortalized ACH-3 cell line is derived from a primary culture of first-trimester normal EVCT isolated from human placenta. The cell lines were cultured in Dulbecco Modified Eagle Medium (DMEM) High Glucose: (#AL007, Himedia) Fetal Bovine Serum (#RM10432, Himedia) and 37°C incubator with humidified atmosphere of 5% CO2 (Heal force, China).
Materials used
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) Reagent (5 mg/ml), Dimethyl Sulfoxide (DMSO), camptothecin, Dulbecco's Phosphate-buffered Saline (DPBS), 96-well plate for culturing the cells (From Corning, USA) and T25 flask (# 12556009, Biolite- Thermo) DMSO, PBN, ACH-3P cell line and ADM22-52 antagonist (Purchased from Sigma Aldrich).
MTT assay
MTT assay is a colorimetric assay used for the determination of cell proliferation and cytotoxicity, based on reduction of the yellow colored water soluble tetrazolium dye MTT to formazan crystals. About 200 μl of cell suspension was loaded in a 96-well plate at required cell density (20,000 cells per well), without the ADM antagonist and allowed the cells to grow for about 12 h. After incubation appropriate concentrations of both positive and negative control were incubated for 24 h at 37°C in a 5% CO2 atmosphere. Later MTT reagent (0.5 mg/ml) was added. Formation of formazan crystals were observed under microscope
Analysis of viability and cell death by Annexin V
The investigated drug, ADM22-52 was applied to cell lines at the following final concentrations: 5, 25, 50, 75 and 100 μM, except for the control cells. The cultures were incubated for 24 h. The half maximal inhibitory concentration (IC50) value of the drug was determined as 36 μM and 62 μM for ACH-3P cells. The cells were analyzed with BD Biosciences Fluorescein Isothiocyanate (FITC) Annexin V Apoptosis Detection Kit I (Technical Data Sheet, Catalog no. 556547). FITC Annexin V staining precedes the loss of membrane integrity which accompanied the latest stages of cell death resulting from either apoptotic or necrotic processes. Therefore, staining with FITC Annexin V is typically used in conjunction with a vital dye such as Propidium Iodide (PI). Culture cells in a 6-well plate at a density of 3 × 105 cells/2 ml and incubated in a CO2 incubator overnight at 37°C for 24 h. Treat the cells with required concentration of ADM22-52 by IC50 as 36 μM and 62 μM. Cells without any drug (Untreated). At the end of the treatment, removed the medium from all the wells into 12 × 75 mm polystyrene tubes and washed with 500 μl PBS. Then added 180 μl of trypsin-Ethylenediaminetetraacetic Acid (EDTA) solution and again incubated at 37°C for 3-4 min. Poured the culture medium back into their respective wells and harvested the cells directly into 12 × 75 mm polystyrene tubes. Centrifuged the tubes for 5 min at 300 X g at 25°C. Carefully decanted the supernatant and re-suspended the cells in 1X binding buffer at a concentration of 1 × 106 cells/ml. Later transferred 100 μl of the solution (1 × 105 cells) to a 5 ml culture tube. Added 5 μl of FITC Annexin V. Gently vortexed the cells and incubated for 15 min at RT (25°C) in the dark. Added 5 μl of PI and 400 μl of 1X binding buffer to each tube and vortexed gently. Analyzed by flow cytometry immediately after addition of PI. FITC (Fluorescein Isothiocyanate) negative and PI negative (Viable normal cells), FITC positive and PI negative (Early apoptotic), FITC positive and PI positive (Late apoptotic), FITC negative and PI positive (necrotic).
DNA strand break labeling with 5-Bromo-2-Deoxyuridine 5-Triphosphate (BrdUT) for analysis by flow cytometry
This method which is often used to detect fragmented DNA utilizes a reaction catalyzed by exogenous Tetrminal Deoxynucleotidyl Transferase (TdT), often referred to as "end-labeling" or "TUNEL" TdT, UTP nick end labeling). The APO-DIRECT™ assay (Cat. No. 6536KK) is a singlestep method for labeling DNA breaks with FITC-dUTP, followed by flow cytometric analysis. In the APO-BRDU™ assay, TdT catalyzes a template independent addition of Br-dUTP to the 3'-hydroxyl (OH) termini of double- and single-stranded DNA. After incorporation, these sites are identified by flow cytometric means by staining the cells with a FITC-labeled anti-BrdUmAb. Later suspend the cells in 1% (w/v) paraformaldehyde in PBS (pH 7.4) at a concentration of 1-2 × 106 cells/ml. Washed the cells in 5 ml of PBS, then pelleted the cells by centrifugation. Repeat the washing and centrifugation and incubated the cells in the DNA labeling solution for 60 min at 37°C. At the end of the incubation time, 1.0 ml of rinse buffer was added to each tube and centrifuged each tube at 300 X g for 5 min and removed the supernatant. Repeated the rinising and centrifugation step again and again. Later re-suspended the cell pellet in 0.5 ml of the PI/RNase staining buffer and analyzed the cells in PI/RNase solution by flow cytometry.
Data analysis
All assays were done in three independent sets of experiments and results were expressed as mean ± SEM. Data of treated cells were compared with their respective controls and were analysed using one way Analysis of Variance (ANOVA) with Student's t-test and determined significance. In all cases P-values of < 0.05 were accepted as statistically significant. All statistical analysis was performed using Graph Pad 3.0 version.
Cytotoxicity assay
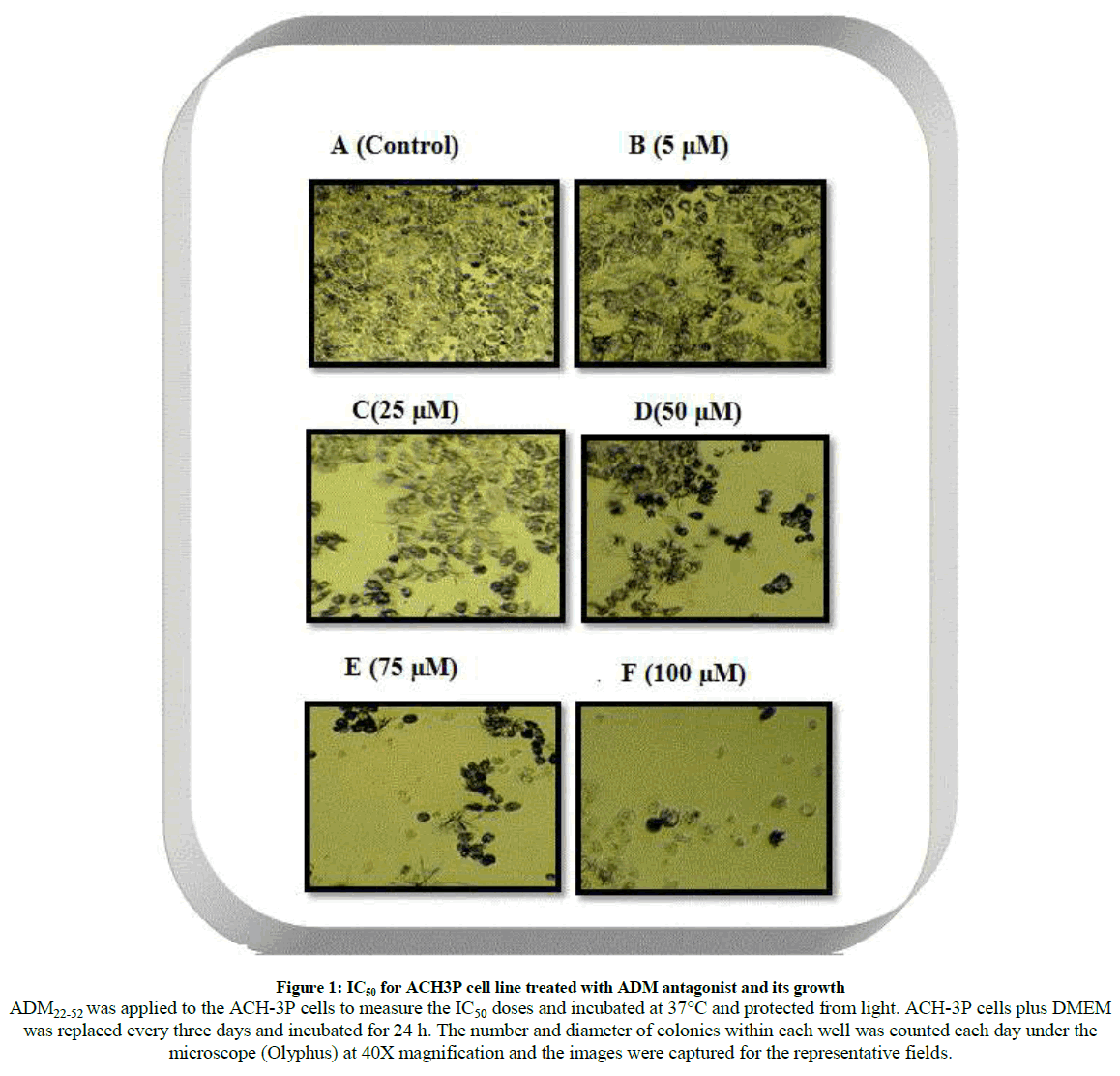
The results for cytotoxicity assay to varying concentration after the exposure to ADM22-52 antagonist (Figure 1) on first trimester cell lines (ACH-3P) whereby the cells displayed alterations in cells formation and morphology of cells become clustered, spherical and they were detached from the surface. The ADM antagonist reduced the cell viability in dose- dependent manner. The number and diameter of colonies within each well was counted each day under the light microscope (Olyphus) and the images were captured for the representative fields.
Figure 1: IC50 for ACH3P cell line treated with ADM antagonist and its growth
ADM22-52 was applied to the ACH-3P cells to measure the IC50 doses and incubated at 37°C and protected from light. ACH-3P cells plus DMEM
was replaced every three days and incubated for 24 h. The number and diameter of colonies within each well was counted each day under the
microscope (Olyphus) at 40X magnification and the images were captured for the representative fields.
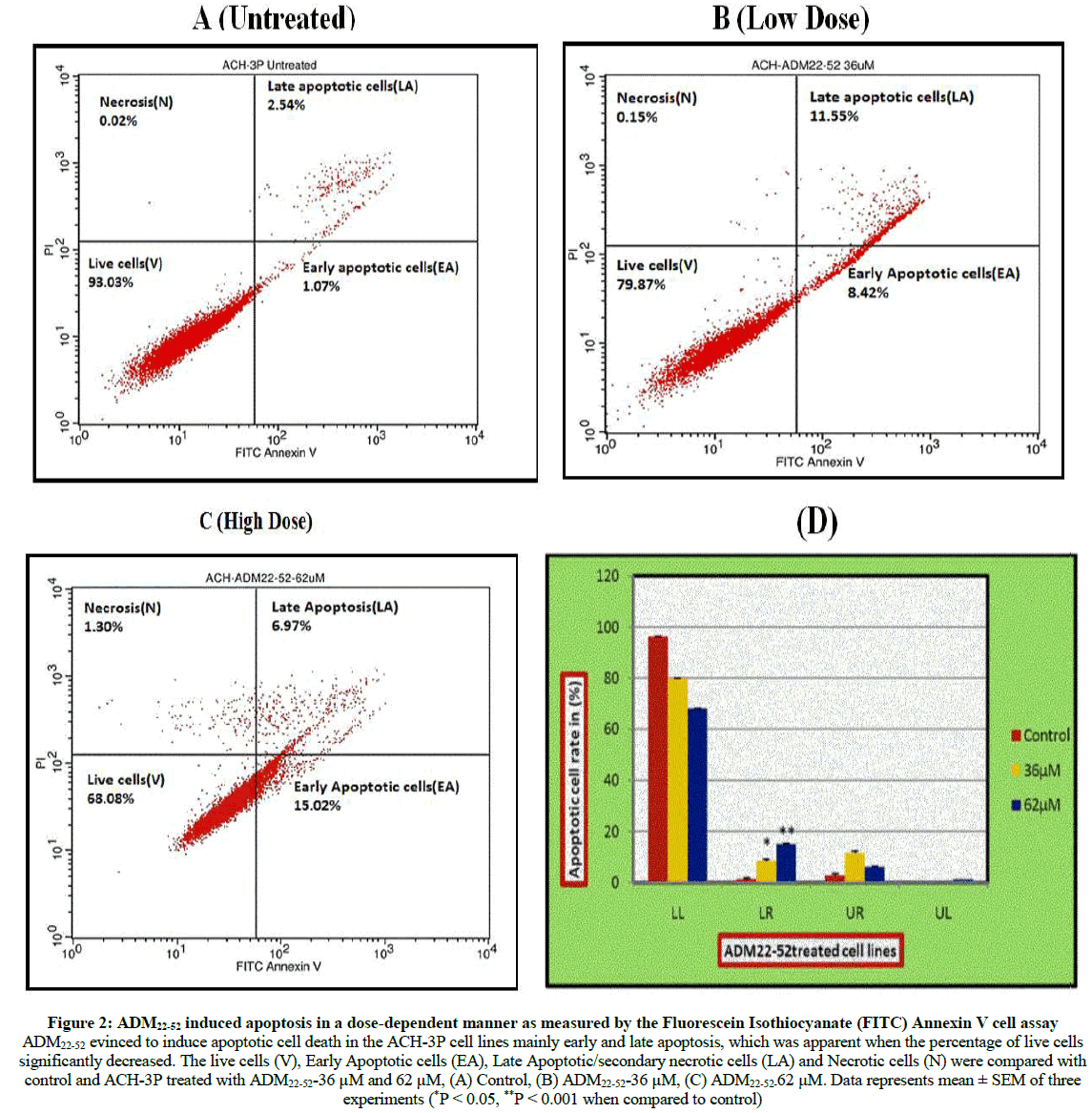
Apoptosis (Annexin V/PI double staining)
The cell population of interest was gated on the basis of untreated control stained cells. These were divided into four quadrants and were analysed as FITC negative and PI negative (Viable normal cells), FITC positive and PI negative (Early apoptotic), FITC positive and PI positive (Late apoptotic) and FITC negative and PI positive (Necrotic). It was observed that there was a decrease in viable cells when compared with control and low dose (36 μM) and high dose (62 μM) (Figure 2a-d).
Figure 2: ADM22-52 induced apoptosis in a dose-dependent manner as measured by the Fluorescein Isothiocyanate (FITC) Annexin V cell assay
ADM22-52 evinced to induce apoptotic cell death in the ACH-3P cell lines mainly early and late apoptosis, which was apparent when the percentage of live cells
significantly decreased. The live cells (V), Early Apoptotic cells (EA), Late Apoptotic/secondary necrotic cells (LA) and Necrotic cells (N) were compared with
control and ACH-3P treated with ADM22-52-36 μM and 62 μM, (A) Control, (B) ADM22-52-36 μM, (C) ADM22-52-62 μM. Data represents mean ± SEM of three
experiments (*P < 0.05, **P < 0.001 when compared to control)
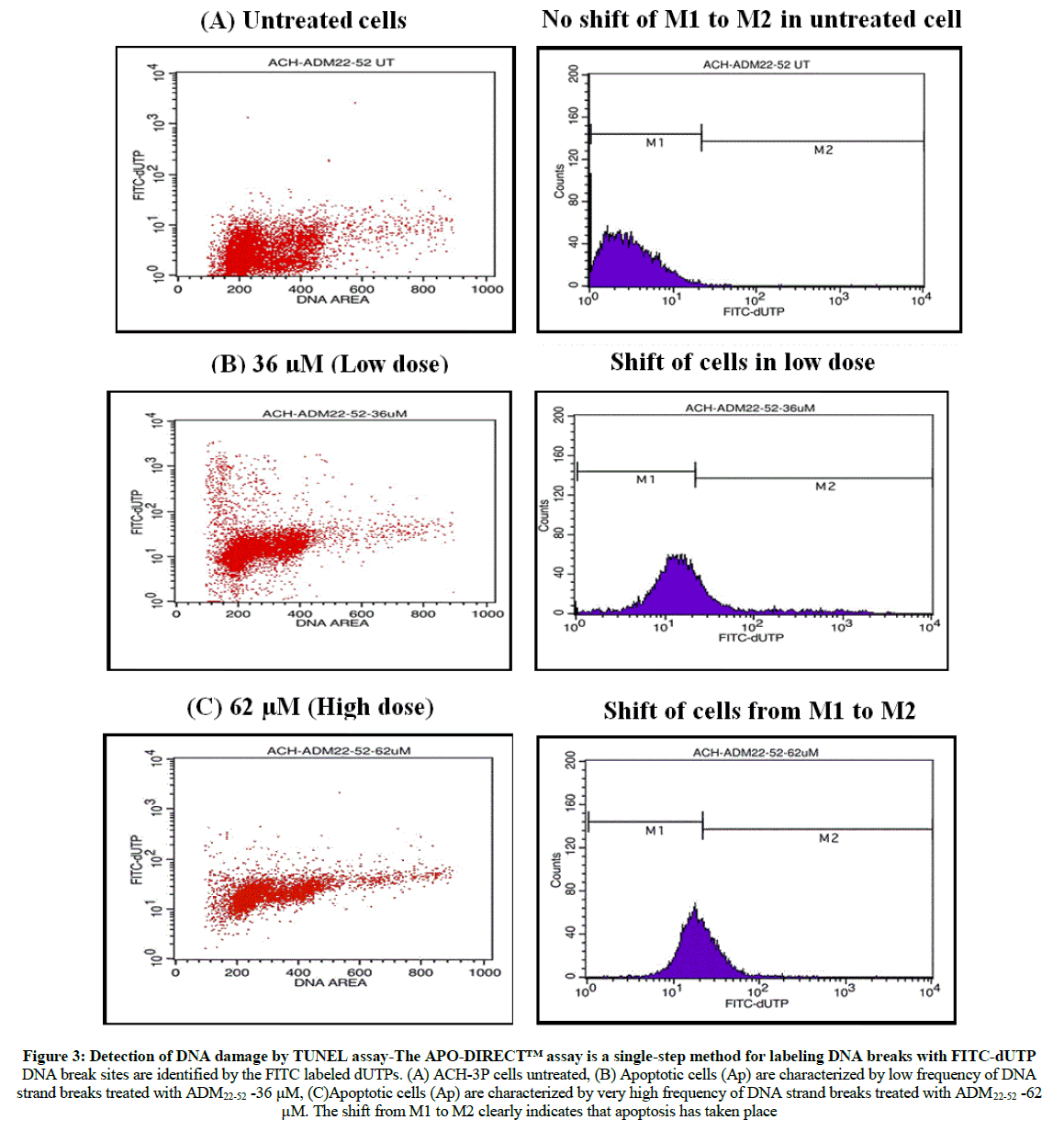
Apoptosis detection by TUNEL Assay using flow cytometry
The TUNEL assay was also performed to further characterize the apoptotic response. The percentage of cells containing DNA strand breaks was quantified by flow cytometry. Using ADM22-52 in ACH-3P cell line which exhibited increase in apoptosis where the dot plots shifted to early apoptotic region on lower right in low dose and in high dose the dot plot shifted to early and late apoptotic region which distinctively signifies that apoptosis has occurred and not necrosis when compared with control (Figure 3).
Figure 3: Detection of DNA damage by TUNEL assay-The APO-DIRECT™ assay is a single-step method for labeling DNA breaks with FITC-dUTP DNA break sites are identified by the FITC labeled dUTPs. (A) ACH-3P cells untreated, (B) Apoptotic cells (Ap) are characterized by low frequency of DNA strand breaks treated with ADM22-52 -36 μM, (C)Apoptotic cells (Ap) are characterized by very high frequency of DNA strand breaks treated with ADM22-52 -62 μM. The shift from M1 to M2 clearly indicates that apoptosis has taken place
Discussion
Implantation in humans begins with invasion of uterine epithelium and underlying stroma by extraembryonic trophoblast cells. Villous cytotrophoblast cells at the tips of some anchoring villi proliferate outward from the underlying basement membrane to form columns from which individual cells migrate into the decidual tissue. These interstitial trophoblast cells invade as far as the superficial layer of the myometrium [17]. ADM and its related peptides CGRP are expressed in uteroimplantation tissues on the endometrium and are reported to be important in maintaining normal pregnancy function [18-22]. ACH-3P are first trimester cell lines which are pure population of first cytotrophoblast cells. ADM has been suggested to be involved in implantation, placentation, embryogenesis and uterine relaxation during pregnancy, as an angiogenic factor, growth enhancing factor, immune modulator and vasodilator reviewed by [23]. As ADM is constitutively released from endothelial cells and its release is regulated entirely at the level of protein expression [24]. The augmentation in ADM plasma levels in disease is associated with an enhancement in ADM gene expression in tissues, especially vascular and smooth muscle cells [25-28].
The present study was undertaken to demonstrate the expression and role of ADM in human placenta cell lines. The assessment of apoptosis is imperative in the evaluation of role of ADM. To measure early apoptotic changes Annexin-V staining and Tunel assay by flow cytometry is reliable. After treating the cell lines with ADM22-52 antagonist, cytotoxicity assessment was done to check the viability of the cells and through graph we plotted IC50 value. The IC50 value for low dose is 36 μM and high dose is 62 μM. By Annexin-V assay we proved that early to late apoptosis has occurred and not necrosis which is shown in bar graph where there is decrease in percentage of viable cell and increase in apoptotic cell.
The TUNEL by flow cytometry analysis is a significant hallmark for apoptosis, where the DNA strand break is labeled with FITC-dUTP. The dot plots shift toward early apoptotic to late apoptosis clearly signifies that apoptosis has taken place. Our studies provide hypothetical evidence of a role for this novel peptide in trophoblast invasion and migration in early human placental function.
Conclusion
During early pregnancy, controlled trophoblast cell invasion into the maternal endometrium is crucial for successful implantation and placentation. Trophoblast cells derived from the trophectoderm, the outermost epithelial cell layer of the blastocyst, initiate entrenchment and implantation into the maternal endometrium through a complex series of interactions between various regulatory factors and an improved understanding of the molecular mechanisms that control trophoblast invasion will help to identify the defects in these processes that contribute to such pregnancy complications as IUGR and preeclampsia. A number of mechanisms have been portrayed in the pathophysiology of pregnancy complications, such as intrauterine growth retardation, gestational hypertension, and preeclampsia. We propose that one such mechanism may be intensified apoptosis caused by decrease in the levels of ADM during pregnancy. There are an assembly of genetic, physiologic, and environmental factors that must all work together in perfect synchronization throughout pregnancy to create the magic so-called “miracle” that is a healthy well growth baby. Any irregularity in this process may result in pregnancy complications, which can include implantation failure, miscarriage, fetal growth restriction, gestational diabetes, Preeclampsia (PE), and preterm birth. Given in this complexity, there is a contemporarily a major interest in ADM and its efforts have been put forth in the field, to expand our understanding of the factors that contribute to healthy versus unhealthy pregnancies.
Acknoweldgement
The authors express their appreciation to Sri Padmavathi Mahila Visvavidyalayam (Women’s) University for providing access to the research facilities and for actively participating in the study and also thanks to the faculty, staff, and students from the Mahila University for their assistance in the research studies. The authors are also highly thankful to Janaradhana P.B. Founder & CEO, Stellixir Biotech, Pvt Ltd., Bangalore, Karnataka. For assisting and guiding in in vitro cell line work and Dr. Penchalaneni Josthna acknowledges the DST-SERB, New Delhi for providing financial assistance.
Funding
These studies were supported by DST-SERB, New Delhi for providing financial support in project by releasing funds timely Project Ref. No.: SB/SO/AS-080/2013.
Ethics approval and consent to participate
Animal studies were performed as per institute animal ethics committee regulations and approved by the committee (Reg. No. 1677/PO/a/12/CPCSEA/SPMVV-IEC/2014).
References
- E. Staun-Ram, E. Shalev, Reprod. Biol. Endocrinol., 2005, 3, 56.
- G.E. Lash, J. Hornbuckle, A. Brunt, M. Kirkley, R.F. Searle, S.C. Robson, J.N. Bulmer, Placenta., 2007, 28, 390-398.
- L. Lunghi, M.E. Ferretti, S. Medici, C. Biondid, F. Vesce, Reprod. Biol. Endocrinol., 2007, 5, 6.
- S.J. NFisher, T.Y. Cui, L. Zhang, L. Hartman, K. Grahl, G.Y. Zhang, J. Tarpey, C.H. Damsky, J. Cell Biol., 1989, 109, 891-902.
- C. Ferretti, L. Bruni, V. Dangles-Marie, A.P. Pecking, D. Bellet, Hum. Reprod. Update., 2007, 13, 121-141.
- P. Bischof, A. Meisser, A. Campana, Placenta., 2000, 21A, S55-S60.
- D. Goldman-Wohl, S. Yagel, Mol. Cell Endocrinol., 2002, 187, 233-238.
- P. Kaufmann, S. Black, B. Huppertz, Biol. Reprod., 2003, 69, 1-7.
- M. Wells, Pathology., 2007, 39, 88-96.
- T. Moriyama, T. Otani, T. Maruo, J. Clin. Endocrinol. Metabol., 2001, 86, 3958-3961.
- S.T.B. Wong, K.K.W. Lam, C.L. Lee, Biol. Reprod., 2013, 88(2), 34, 1-11.
- M. Garayoa, E. Bodegas, F. Cuttittad, L.M. Montuenga, Microsc. Res. Tech., 2002, 57, 40-54.
- J. Penchalaneni, S.J. Wimalawansa, C. Yallampalli, Biol. Reprod., 2004, 71, 1475-1483.
- U. Hiden, C. Wadsack, N. Prutsch, M. Gauster, U. Weiss, H.G. Frank, U. Schmitz, C. Fast-Hirsch, M. Hengstschla¨ger, A. Po¨tgens, BMC Dev. Biol., 2007, 7, 137-150.
- J.L. Janneau, J. Maldonado-Estrada, G. Tachdjian, I. Miran, N. Motte, P. Saulnier, J.C. Sabourin, J.F. Cote, B. Simon, R. Frydman, G. Chaouat, D. Bellet, J. Clin. Endocrinol. Metab.,2002, 87, 5336-5339.
- N. Wolf, W. Yang, C.E. Dunk, I. Gashaw, S.J. Lye, T. Ring, M. Schmidt, E. Winterhager, A. Gellhaus, Endocrinol.,2010, 151, 2835-2845.
- T.D. Burrows, A. King, Y.W. Loke, Hum. Reprod. Update., 1996, 2, 307-321.
- J.C. Stevenson, D.W.R. MacDonald, R.C. Warren, M.W. Booker, BMJ., 1986, 293, 1329-1330.
- R. Di Iorio, E. Marinoni, E.V. Cosmi, Gynecol. Endocrinol., 1998, 12, 1-6.
- R. Di Iorio, E. Marinoni, C. Letizia, B. Villaccio, A. Alberini, E.V. Cosmi, Eur. J. Endocrinol., 1999, 140, 201-206.
- K.K.T. Kobayashi, T. Aso, Y. Hirata, T.M.F. Imai, Eur. J. Endocrinol., 2000, 142, 683-687.
- Y.L. Dong, S. Vegiraju, M. Chauhan, C. Yallampalli, Mol. Hum. Reprod., 2003, 9, 481-490.
- C. Wilson, L.L. Nikitenko, I.L. Sargent, M.C. Rees, Angiogenes., 2004, 7, 203-212.
- S. Sugo, N. Minamino, K. Kangawa, K. Miyamoto, K. Kitamura, J. Sakata, T. Eto, H. Matsuo, Biochem. Biophys. Res. Commun., 1994, 201, 1160-1166.
- T. Ishihara, J. Kato, K. Kitamura, F. Katoh, S. Fujimoto, K. Kangawa, T. Eto, Life Sci., 1997, 60, 1763-1769.
- S. Sugo, N. Minamino, H. Shoji, K. Kangawa, K. Kitamura, T. Eto, H. Matsuo, Biochem. Biophys. Res. Commun., 1994, 203, 719-726.
- S. Sugo, N. Minamino, H. Shoji, K. Kangawa, K. Kitamura, T. Eto, Biochem. Biophys. Res. Commun., 1995, 207, 25-32.
- P.M. Lenhart, K.M. Caron, Trends Endocrinol. Metabol., 2012, 23(10), 524-532.