Research Article - Der Pharma Chemica ( 2017) Volume 9, Issue 3
A Validation of RP-HPLC Method for Determination of Deoxyelephantopin and Isodeoxyelephantopin from Elephantopus scaber L.
Deddi P Putra1*, Yossi Fitrianti2 and Amri Bakhtiar12National Agency Drug and Food Control, Pekanbaru, Indonesia
Deddi P Putra, Faculty of Pharmacy, Andalas University, Padang, Indonesia, Email: putra_aries64@yahoo.com
Abstract
A reversed-phase high-performance liquid of chromatography (RP-HPLC) method for determination of two isomer sesquiterpene lactone deoxyelephantopin (1) and isodeoxyelephantopin (2) from Elephantopus scaber L. was developed and validated. Separation was achieved on a Phenomenex Luna C-18 (250 × 4.6 mm, 5 μm) column, with water: acetonitrile: 2-propanol (66:20:14, v/v/v) as mobile phase, flow rate of 1.0 mL-1 and UV detection was set up at 210 nm using DAD detector. Linearity for both compounds was found in the concentration range of 0.251-1.506 and 0.516-3.096 μg mL-1 (r2 ≥ 0.99), LOD is 0.094 and 0.151 μg mL-1, LOQ is 0.285 and 0.457 μg mL-1, and the range of recovery is 97.64-104.98% and 95.23-102.25%, respectively. The results of intra- and inter-day validation (n=3) showed the method was efficient and could be applied for the determination of sesquiterpene lactones in Elephantopus scaber L. extract.
Keywords
Elephantopus scaber L., Deoxyelephantopin, Isodeoxyelephantopin, RP-HPLC
Introduction
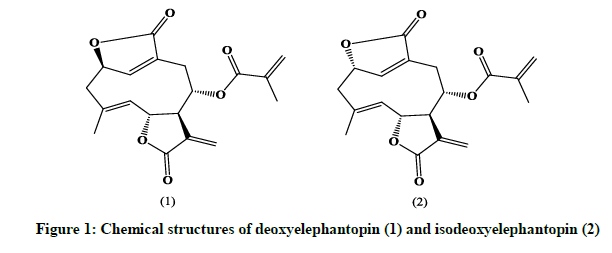
Elephantopus scaber L. (Fam. Asteraceae) is a small herb found in Neotropics, Europe, Asia, Africa and Australia [1]. The plant parts of this herb have been used traditionally for the treatment of a number of diseases in many countries. The plant has been extensively screened for anticancer activity [2-5]. Sesquiterpene lactones such as deoxyelephantopin (1) and isodeoxyelephantopin (2) have been found to be prominent anticancer constituents [2]. Therefore, the analysis of this compound is a great significance to evaluate the quality of this medicinal plant [6]. Until now there has been no technique was published to the simultaneous analysis deoxyelephantopin and isodeoxyelephantopin. In this study, a simple and feasible method has been developed for the simultaneous quantification of two bioactive sesquiterpene lactones from Elephantopus scaber L. and for the quality control of this well-known herbal medicine. MATERIALS AND METHODS Reagents and chemicals Deoxyelephantopin (1) and Isodeoxyelephantopin (2) (Figure 1) used as standard and methanol extract of Elephantopus scaber L. leaves were a gift from Laboratory Biota of Sumatera, Faculty of Pharmacy, Andalas University.
HPLC grade acetonitrile and methanol, analytical grade 2-propanol (Merck, Germany) were used for the HPLC mobile phase. Ultrapure water was used obtained from a Merck Millipore water purification system. Standards and extract were diluted using a mixture of acetonitrile-water.
Equipment
A Shimadzu liquid chromatography system (Shimadzu LC 20 AD) equipped with a quaternary solvent delivery system, an autoinjector and a DAD detector, was used for quantitative analysis. The quantitative analysis was carried out on a Phenomenex Luna C18 column (250 × 4.6 mm, 5 μm). The mobile phase consists of water, acetonitrile and 2-propanol (66:20:14, v/v/v). UV absorption was monitored at 210 nm. The flow rate was 1.0 ml min-1 and sample injection volume was 20 μl.
Preparation of standard and sample solutions
Accurately weighed each standard solution was prepared by dissolving the reference standards in water: acetonitrile (66:34, v/v) to a final concentration of 2.50 μg mL-1 for deoxyelephantopin (1) and 5.00 μg mL-1 for isodeoxyelephantopin (2). This solution was further diluted to appropriate concentrations for establishing calibration curves. The Limit of Detection (LOD) and Limit of Quantification (LOQ) values calculated from calibration curves. For recovery assay, 100 mg methanol extract of Elephantopus scaber L. leaves was transferred into a 50 ml volumetric flask adjusted with water : acetonitrile (66: 34, v/v) and sonicated for 15 min, filtered through a 0.45 μm membrane and added with three difference concentration of deoxyelephantopin standard before HPLC analysis. While for isodeoxyelephantopin recovery assay, the extract used is 40 mg and prepared in the same condition.
Method Validation
Specificity
Standard solution and methanol extract of Elephantopus scaber leaves were injected separately to confirm the retention times. Deoxyelephantopin and isodeoxyelephantopin peaks were eluted at 13.869 and 14.751 min, respectively. The resolution between the peaks was found to be 1.985.
Linearity
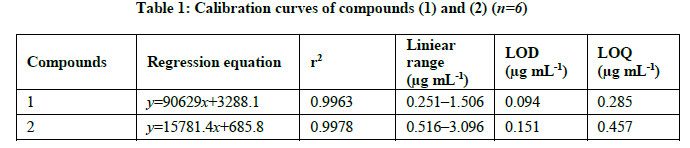
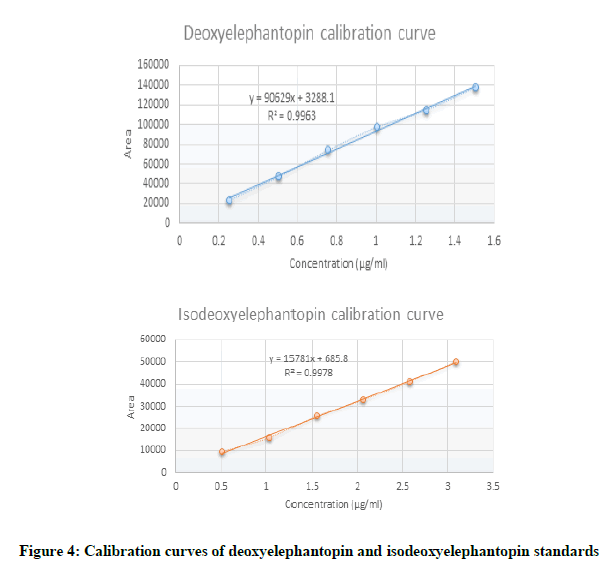
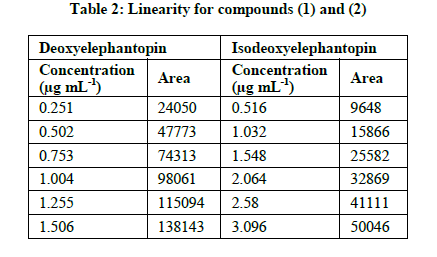
Deoxyelephantopin standard solutions at six different concentration levels ranging from 0.25, 0.5, 0.75, 1.0, 1.25 and 1.5 μg mL-1 were prepared. Isoeoxyelephantopin standard solutions at six different concentration levels ranging from 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 μg/mL were prepared. Each samples standard solution was injected in duplicate. The mean responses recorded were plotted against concentration. The correlation coefficient for deoxyelephantopin was found to be 0.9963 and calibration equation was found to be y=90629x+3288.1. The correlation coefficient for isodeoxyelephantopin was found to be 0.9978 and calibration equation was found to be y=15781x+685.8. The linearity, regression, and linear ranges of the HPLC method were determined (Table 1). The correlation coefficient (r2 ≥ 0,996) values indicated good correlations between concentrations of investigated compounds and their peak areas within the test ranges (Figure 4).
Limit of detection (LOD) and limits of quantification (LOQ)
The limit of detection and limit of quantification for deoxyelephantopin and isodeoxyelephantopin were calculated from the linearity data using residual standard deviation of the response and slope of the calibration curve. LOD and LOQ values for deoxyelephantopin were less than 0.094 and 0.285 μg mL-1, respectively. The LOD and LOQ values for isodeoxyelephantopin were less than 0.151 and 0.457 μg mL-1, respectively (Table 2).
Accuracy
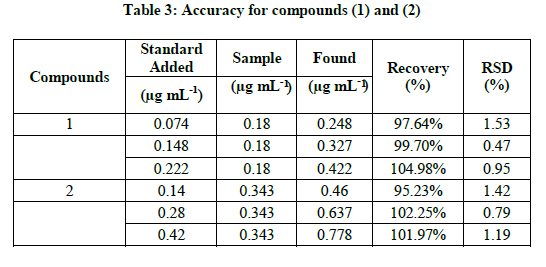
Methanol extract of Elephantopus scaber L. leaves were spiked with deoxyelephantopin at 0.074, 0.148 and 0.222 μg mL-1 and isodeoxyelephantopin standard at 0.140, 0.280 and 0.420 μg mL-1. Each spiked solution was prepared in triplicate and injected. The recovery percentage and % RSD were calculated. The developed method has a good accuracy with the overall recovery of 95.23% to 104.98% (Table 3). The acceptance criteria for recovery at each level were between 80 and 120% as per in-house validation protocol [7].
Precision
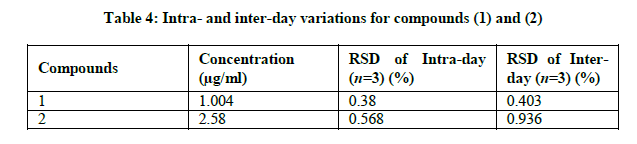
Repeatability (intra-day) was demonstrated by analyzing standard on three different time in one day. Intermediate precision (interday) was demonstrated by analyzing standard on three different day. As shown in Table 4, the HPLC method has good reproducibility for the quantification of the two sesquiterpene lactones; deoxyelephantopin with intra- and inter-day variations less than 0.380 and 0.403% respectively and isodeoxyelephantopin with intra- and inter-day variations less than 0.568 and 0.936% respectively.
These results indicated that the HPLC method is precise, accurate, and sensitive for quantitative analysis of deoxyelephantopin and isodeoxyelephantopin in Elephantopus scaber L. sample.
Conclusion
A simple, rapid and accurate HPLC method was developed for the determination of bioactive constituents in Elephantopus scaber L. This was the first report on the simultaneous quantification of the two isomer sesquiterpene lactones deoxyelephantopin and isodeoxyelephantopin from Elephantopus scaber L. Our results suggest that this method could be utilised for routine analysis for sample containing Elephantopus scaber L. extract.
Acknowledgements
We are thankful to PUPT-DP2M research funding achieved by DPP, to Badan POM RI for YF magister scholarship, and to Laboratory Biota of Sumatra Andalas University as well as BBPOM of Pekanbaru for facilities and HPLC instruments.
References
[1] C.A. Backer, B. Van Den Brink, Flora of Java., 1965, II.
[2] S.M. Hiradeve, V.D. Rangari, Elephantopus Scaber Linn. Nat. Prod. Res., 2014, 28, 11, 819-830.
[3] S.M. Hiradeve, V.D. Rangari, J. Appl. Biomed., 2014, 12, 49-61.
[4] B.S. Geetha, M.S. Nair, P.G. Latha, P. Remani, In. Vitro. J. Biomed. Biotechnol., 2011, 1-8.
[5] F.A. Kabeer, R. Prathapan, Pharmacologia., 2014, 5, 272-285.
[6] International Conference on Harmonisation. Q2(R1), 2005.
[7] Association of Official Analytical Chemist. Guidelines for Standard Method Performance Requirements, 2012.



